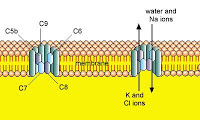
 Eculizumab is a humanized monoclonal antibody that binds to and prevents activation of the C5 complement protein. As you may remember, this protein is critical to the formation of the membrane attack complex (MAC) in complement dependent immune activation.
Eculizumab is a humanized monoclonal antibody that binds to and prevents activation of the C5 complement protein. As you may remember, this protein is critical to the formation of the membrane attack complex (MAC) in complement dependent immune activation. Eculizumab has been licensed for the treatment of paroxysmal nocturnal haemoglobinuria (PNH), and has proven efficacy in reducing intravascular haemolysis, reducing transfusion requirements and stabilizing haemoglobin concentrations. However, it is very expensive and may require life-long treatment in these patients, as it does not alter the underlying cause of PNH.
It has been used off label in the renal transplantation setting for the treatment of refractory humoral rejection. An interesting case report highlighted it’s use. Severe antibody-mediated rejection (AMR) occurred following a living related renal transplant in a highly sensitized patient, who required pre-transplant plasmapheresis and IVIg for the presence of preformed donor specific antigens (DSA). Flow crossmatch was negative at time of transplantation, but became positive again on day 6 due to DSA against the donor A2 antigen, necessitating further plasmapheresis and IVIg. Renal function deteriorated and biopsy was consistent with severe AMR.
On day 10 he was given eculizumab and rituximab, with a temporary hold on plasmapheresis to prevent removal of the administered drugs. Within 48 hours his urine output had increased and by five days his creatinine began to decline. Plasmapheresis and IVIg were restarted in order to deal with the persistent DSA. Although it is impossible to ascribe the recovery solely to eculizumab, a follow-up biopsy illustrated markedly decreased staining for the MAC, at least supporting the efficacy of eculizumab in reduction of inflammation.
Due to the central role of the complement system in fighting infection, it is recommended that patients are given the N. meningiditis vaccine prior to treatment with eculizumab. This novel drug may have a potential role in the treatment of resistant humoral rejection in the future, but a major limiting factor is the cost. There is little long-term safety data in the treatment of immunosuppressed patients with eculizumab at this point in time.


Another nephrological therapeutic indication for eculizumab…
There is a growing number of case reports suggesting that eculizumab may be effective also in the treatment of atypical hemolytic uremic syndrome, mainly in those pts with factor H mutations.