A gentleman with ESRD secondary to polycystic kidney disease comes in with vomiting and headache eight months after kidney transplantation. Brain MRI shows a small single enhancing lesion in the temporal lobe. Lumbar puncture reveals an elevated opening pressure (41), WBC 121 (77% lymphs), elevated total protein (210) and low glucose (41). Cultures are negative.
The top 3 differentials of CNS masses in post-transplant kidney recipients are: neurotoxoplasmosis, lymphoma and tuberculoma. EBV negative status prior to transplantation increases your risk of post-transplant lymphoproliferative disorder (PTLD) and checking EBV VL could help with initial evaluation. Coming from endemic areas of tuberculosis (TB) raises the suspicion for tuberculosis. TB in kidney transplant recipients is a particular challenge both diagnostically and therapeutically. Atypical clinical presentations (extra-pulmonary) and negative tuberculin skin tests/sputum smears are frequent. The treatment of TB also has its own challenges, which include pharmacokinetic interactions between immunosuppressive and antituberculous medications (reduction of FK by more than 60% is required), allograft-related drug toxicities, and inadequate immune responses to Mycobacterium tuberculosis due to exogenous immunosuppression.
In general, empirical therapy of neurotoxoplasmosis is instituted in immunosuppressed patients with an enhancing brain lesion, consisting of sulfadiazine, pyrimethamine and leucovorin. Reduction of immunosuppression is also indicated and follow up imaging is performed. Last resource if no improvement and negative work up is to perform a brain biopsy to obtain tissue specimen.
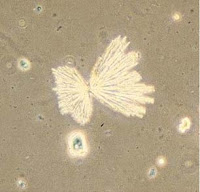
Treatment of neurotoxo with high doses of sulfadiazine may precipitate acute kidney injury by crystal formation (~30% of patients), in particular if volume depletion is present and urine pH is 5.5 or less. Intra-renal crystalluria may be prevented by maintaining fluid intake above three liters per day (PO or IV). Monitoring of serial urinalyses for the development of crystalluria is recommended. For those developing crystalluria, intravenous bicarbonate solution to alkalinize the urine (goal pH above 7.15) is administered. The patient above developed significant AKI (creatinine increased from 1 to 4) after sulfadiazine treatment and crystals were identified in the urine (representative figure).
Be aware of potential drugs/toxins** that might form intratubular crystals and precipiate AKI. Act pre-emptively with fluid hydration +/- alkalinization of urine depending on the toxin.
** Acyclovir (IV), sulfonamides, ethylene glycol, high dose vitamin C, methotrexate, protease inhibitors, phophate enemas, orlistat
*** Remember that for MTX renal toxicity, there is glucarpidase as a last resource to quickly reduce MTX levels in patients with AKI and persistently high MTX levels 48 hrs after dosing.