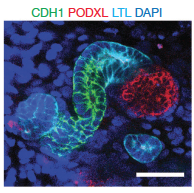
Two papers demonstrated the feasibility of developing kidney cells from human pluripotent stem cells. One from the USA by Dr Joseph Bonventre’s group at the Renal Division of the Brigham and Women’s Hospital (Morizane, Lam et al. Nat Biotechnol 2015) and one from Australia by Dr Melissa Little’s Group at University of Melbourne (Takasato et al. Nature 2015).
Albert Lam, one of the co-authors of the Nat Biotechnol paper, stated that: “We established a differentiation protocol that would mimic the stages of nephron formation as closely as possible. Our approach in recapitulating the steps of kidney development resulted in a highly efficient protocol for generating kidney organoids. The ability to do this using induced pluripotent stem (iPS) cells, which are derived from skin or blood cells of patients, allows the creation of kidney tissue without ethical concerns and allows the tissue to be “personalized”, that is, generated from a particular patient. If in the future the tissue is re-implanted back into the patient, the immune response may then be very limited since the tissue will be recognized as self.”
This achievement has an enormous potential impact on a variety of clinical and translational applications, including kidney tissue bioengineering to replace lost kidney tissue, renal assist devices to treat acute and chronic kidney injury, drug toxicity screening, screening for novel therapeutic agents, and human kidney disease modeling.
Albert kindly shared with me that I shouldn’t be anxious yet about my job as a transplant nephrologist since more years are needed to develop a full organ incorporating the blood supply and urinary collecting system. I selfishly hope so…