 The May issue of AJKD (Vol.55, No.5) had a nice supplement on TLS and AKI. Here is the link.
The May issue of AJKD (Vol.55, No.5) had a nice supplement on TLS and AKI. Here is the link.
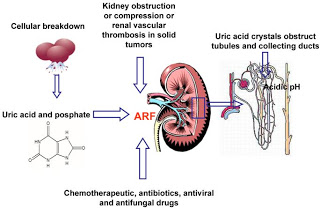
Tumor lysis syndrome (TLS) describes a condition with significant clinical and lab abnormalities caused by rapid and massive tumor cell death. Occurring either spontaneously or after chemotherapy, TLS is a medical emergency and is associated with significant morbidity and if untreated mortality.
The risk for patients of developing TLS changes with their type of malignancy:
2. Intermediate risk: Diffuse large B-cell lymphoma, acute ALL (WBC 10-50K), acute AML (WBC 10-50K), CLL (WBC 10-100K, treated with fludarabine), malignancies with rapid proliferation with expected rapid response to therapy
3. Low risk: indolent NHL, acute ALL with WBC
Definition of TLS is as follows:
1. Definition of Laboratory TLS (any 2 or more criteria within 3 days before or 7 days after chemotherapy):
– Uric Acid > 8mg/dl or 25% increase from baseline
– Potassium > 5 mEq/L or 25% increase from baseline
– Phosphorus > 6.5 mg/dl (children) or > 4.5mg/dl (adults) or 25% increase from baseline
– Calcium less than 7mg/dl or 25% decrease from baseline
2. Definition of clinical TLS (laboratory TLS plus at least one of the below criteria):
– Creatinine greater than 1.5 value of upper limit of age-adjusted normal range
– Cardiac arrhythmias or sudden death
– Seizure
Acute Kidney Injury in TLS occurs mainly through crystal deposition including
a) uric acid
b) calcium phosphate
Other aggravating factors include:
c) volume depletion / hypotension / CHF
d) extrinsic urinary obstruction
e) pre-existing CKD
f) nephrotoxic medications such as NSAID
g) radiocontrast exposure
h) sepsis
Prevention and treatment of the AKI caused by TLS focuses on reversing above factors, requires volume depletion, and treatment of electrolyte abnormalities (hyperkalemia, hyperphosphatemia). The primary goal is to minimize or treat hyperuricemia by either:
Decreasing production of Uric acid with Allopurinol, which blocks Xanthin oxidase,
and/or
Converting Uric acid into water-soluble Allantoin with Rasburicase (Urate oxidase)
Of these approved treatments for hyperuricemia, the former with allopurinol can lead to Uric acid independent crystal deposition by accumulation of Xanthin products. In addition, elevated urinary Uric acid levels may lead to crystal-independent AKI by by alteration of renal autoregulation and vasoconstriction. Routine urinary alkalinization to prevent Uric acid crystals is controversial since it may increase risk for renal calcium phosphate crystal deposition. Dialysis therapy should be initiated when indicated using standard criteria for AKI.



I think a stronger statement can be made about alkalinization than to simply say it is controversial — in the Onc world, alkalinization is now frankly advised against due to the risk of calcium phosphate precipitation (see, for example, J Clin Oncol 26:2767-2778).
Yes, prophylactic rasburicase prior to chemo. The oncology group gives it for high risk cases (bulky lymphoma, high blast count leukemia, prior TLS).
@ Graham:
Are these folks with undetectable levels of uric acid getting prophylactic rasburicase? Or do they initially have hyperuricemia and then resolution prior to any AKI? Just wondering what the indication for rasburicase was in these settings. We only use it at my institution if there's clearly hyperuricemia with the TLS.
Given the frequency with which rasburicase is now used I wonder if there is a shift in presentation towards more calcium phosphate mediated injury?
I was always taught that urate nephropathy was the primary cause of AKI in tumor lysis syndrome but the last three cases I've seen have all had undetectable uric acid levels.
Food for thought.