In our dialysis unit, the standard HCO3 bath is 35 mmol/L. While it is adjustable, it is rarely changed but recently we had a patient with a pre-dialysis HCO3 of 12 which got me thinking about what constitutes an abnormal serum HCO3 in a dialysis patient. Under normal circumstances, your serum HCO3 level is determined by alkali regeneration in the kidney but in dialysis patients this changes to alkali addition across the filter. As a result, there are significant swings in HCO3 levels across the week ranging from 26-30 mmol/L post-dialysis to 20-24 mmol/L pre-dialysis. The pre-dialysis HCO3 is determined by:
– End dialysis HCO3 – Rate of production of endogenous acids (which correlates closely with protein ingestion) – Time between dialysis sessions – Free water retention between sessions (HCO3 falls by 0.5 mmol/L for each liter of free water retained) – Bicarbonate losses (urine/diarrhea)
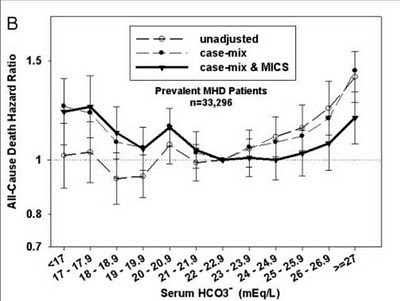
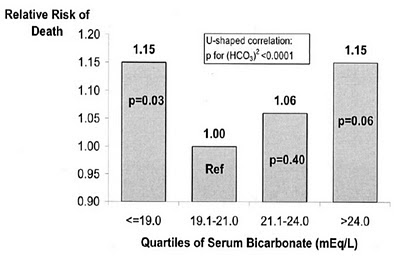
The KDOQI guidelines suggest that pre-dialysis HCO3 should be around 22 but what is the evidence for this? There are two major studies which can guide us. The first is the DOPPS study where the authors took 7000 patients dialysis patients and found that there was a U-shaped curve of RR of death. Those with a HCO3 of less than 17 and greater than 24 were at an increased risk of death. However, when adjusted for nutritional factors and co-morbidities, a high HCO3 was no longer independently associated with mortality while a HCO3 of less than 17 remained significant.
In the second study the authors looked at 36000 dialysis patients and again found that a HCO3 of less than 17 was independently associated with mortalilty while the increased mortality seen in those with a HCO3 of greater than 24 could be entirely explained by nutritional status and co-morbidities.
Despite all of these data, neither of these studies address the issue of whether correcting acidosis in these patients influences outcome. There are a few small randomized trials in hemodialysis patients which show some benefit but are too small to really answer this question. There have, of course, been a number of studies in patients with CKD demonstrating that correction of acidosis improves outcomes. Probably the best advice at this time is that if you come across a patient with a HCO3 of greater than 24, you should assess them for the presence of an acute acid base disorder and then assess their nutritional status. In a patient with an unusually low HCO3, ensure that delivery of HCO3 during dialysis is adequate (by measuring post-dialysis HCO3), determine if HCO3-wasting is present and then increase the HCO3 bath to maintain the HCO3 greater than 18.
This is a great review on the subject published earlier this year.

That said, then why are KDOQI guidelines still around 22 for pre-ESRD (rather than 24)
It seems like this would essentially amount to accepting a lower standard in the CKD population.
You must remember that the bicarb level that we use as a reference in dialysis patients is immediately pre-dialysis and as a result is the lowest level that they are exposed to during their treatments. Their average bicarbonate levels are much higher (in the 22-24 range) and as a result are comparable with the levels seen in the CKD studies.
Interesting. Recent "bicarbonate supplementation" studies such as Ashurst, Mahajan ranging from CKD stage II-IV+ are suggesting serum bicarbonate around 24 (post intervention) has lower risk factor in the pre-ESRD population. Very small studies, but consistent with recent much larger associational type studies (Shah 2009, Raphael 2010)also suggesting that higher than 22 (24-26 range typical) seems superior in non-dialysis population.
Why might a higher serum bicarbonate level (whether intrinsic, or achieved through intervention) be seen for this non-dialysis population to be associated with better outcomes? (Perhaps the same curve, but shifted slightly.)
That is an excellent question. His urine pH was 8.0 both pre and post dialysis and he had a urinary anion gap of +39. Our presumptive diagnosis is a proximal RTA combined with an inability to excrete ammonium ions which may be secondary to a distal RTA or just his underlying renal disease. He has a urine output of 3L daily and his renal diagnosis is chronic urinary tract obstruction which has been found to be associated with the hyperkalemic variant of a distal RTA. Still haven't figured it out completely but it has implications for his management post-transplant
I would like to know what was the reason for the severe metabolic acidosis? This is clearly not normal and indicates either a significant exogenous or endogenous acid load, or significant HCO3 loss (which could only be GI). DIlution to this degree is essentially impossible. THe DD is actually very limited but the case very interesting.
A special issue: HCO3 serum concentration cycling in manteinance HD pts. Any study on this topic?