In continuous renal replacement therapy (CRRT), the coagulation cascade is activated due to blood contact with the extracorporeal circuit. Therefore, anticoagulation is often required during CRRT to prevent premature clotting and maintain circuit patency. Unfractionated heparin has been the standard choice in the past owing to its efficacy and low cost. However, because of the relatively high incidence of bleeding complications associated with systemic anticoagulation, regional anticoagulation of the circuit with citrate has emerged as an attractive alternative.
Citrate is an organic acid with a low molecular weight (298 daltons). By chelating calcium, sodium citrate inhibits thrombin generation and prevents coagulation.
While there are numerous protocols for regional citrate anticoagulation that differ by the solution type and CRRT modality, in general, the citrate infusion rate is titrated to maintain the ionized calcium concentration in the extracorporeal circuit (measured as the post-filter ionized calcium) less than 0.35 mmol/L to facilitate adequate anticoagulation.This corresponds to a blood citrate concentration of approximately 4-6 mmol/L in the circuit. Due to the risk of hypocalcemia, calcium chloride or calcium gluconate solution should be administered either at the end of the circuit or through a separate central line. Postfilter, systemic and total calcium levels should be monitored regularly (every 6 hs). The majority of the calcium-citrate complex is removed across the hemofilter and the remaining amount, infused into the patient, is rapidly metabolized to bicarbonate via the citric acid (Krebs) cycle, in the liver, muscle, and kidney cortex. Under normal conditions, citrate’s half-life is approximately 5 minutes. A schematic of the CRRT circuit with various elements of the regional citrate anticoagulation is shown in Figure 1.
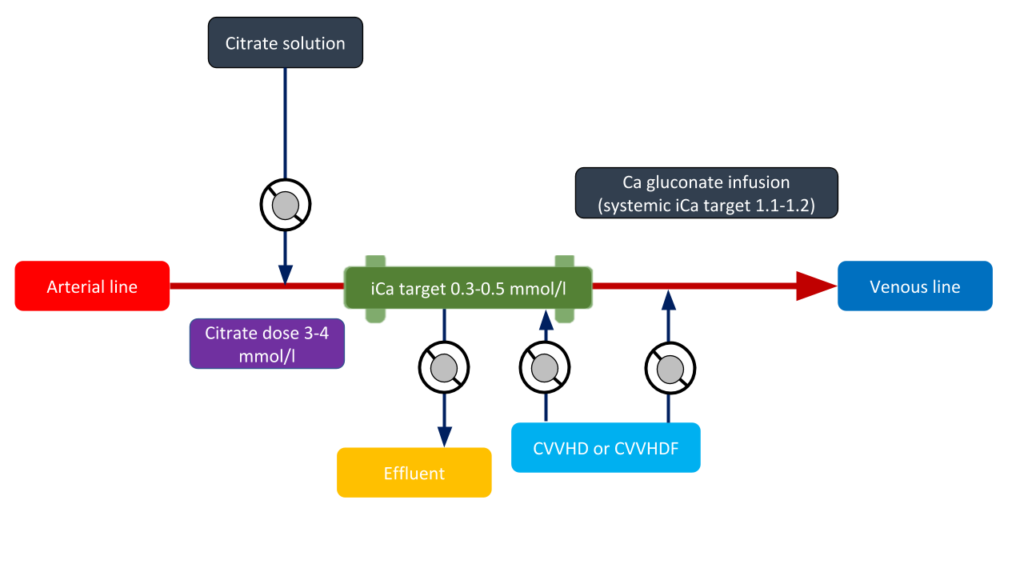
Regional citrate anticoagulation (RCA) vs Heparin: In a randomized controlled trial comparing the efficacy and safety profile of RCA and systemic heparin, Stucker et al observed the effective delivered daily RRT doses were 29 ± 3 and 27 ± 5 mL/kg/hr in the RCA and heparin groups, respectively. Interestingly, the filter lifespan was significantly better at 49 ± 29 hrs in the RCA group compared to 28 ± 23 hrs in the heparin group, though patient survival rates at 28 and 90 days did not differ. In a meta-analysis of 6 randomized controlled trials including 488 patients, RCA was associated with a significant decrease in bleeding complications compared to heparin (RR, 0.34; 95% CI, 0.17-0.65) but with major hypocalcemia events. In terms of the necessity for blood transfusion, Morabito et al demonstrated a lower transfusion rate during RCA-CVVH than with heparin (0.29 vs. 0.62 blood units/day, p = 0.017) or no-anticoagulation (0.29 vs. 0.64 blood units/day, p = 0.019) in addition to lower need for filter replacement during RCA-CVVH in a cohort of patients undergoing cardiac surgery.
In addition to the above studies using unfractionated heparin, RCA has been compared to systemic anticoagulation with low-molecular weight heparin nadroparin in a randomized controlled trial, which demonstrated survival benefit with RCA. RCA-CVVH reduced 3-month mortality in patients post surgery, with sepsis, with higher sepsis-related organ failure assessment (SOFA) scores, and those with lower age. The RICH trial is an ongoing prospective, randomized, multicenter study investigating the effect of RCA on filter life span and 90-day overall survival. It is expected to provide more definitive answers regarding the appropriate choice of anticoagulation. The KDIGO guidelines recommend using RCA as the preferred modality for CRRT in centers where it is available unless the patient has a contraindication.
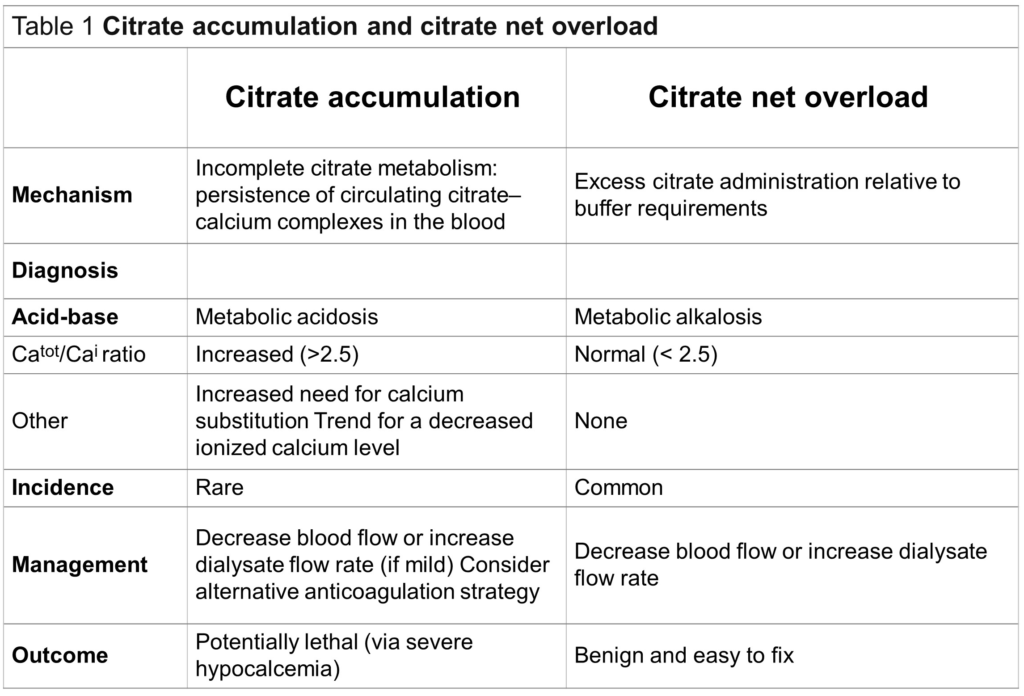
Metabolic effects of RCA: Citrate solutions have a high sodium content (3 Na+ per one citrate molecule in trisodium citrate) causing plasma alkalinization due to an increase in the plasma strong ion difference under normal conditions. However, because citrate is a weak acid, when its catabolism is impaired the calcium-citrate complex accumulates and generates acidosis. In this context, it is important to differentiate between citrate accumulation and citrate net overload. Citrate accumulation occurs when the citrate administration exceeds the body’s capacity to metabolize it (e.g. severe liver failure) and therefore, calcium-citrate complex remains in the blood. To detect the citrate accumulation we use the total/ionized calcium (Ca/Ca++) ratio, with a value equal or greater than 2.5 indicates continuous accumulation of citrate. Total calcium here reflects the calcium bound to citrate.The decrease in ionized calcium can lead to potentially life-threatening complications. Note that to calculate this ratio,both the numerator and the denominator must have the same units, that is, total calcium value must be in mmol/L. The conversion factor is approximately 0.25, which means 10 mg/dL of calcium is equal to 2.5 mmol/L.
On the other hand, the overload of citrate and sodium that occurs in cases of excessive citrate administration or low clearance in the hemofilter lead to plasma alkalinization. This situation is easy to fix by decreasing the blood flow or increasing the dialysate or replacement fluid rate.
With closely monitoring of the acid-base status and serum electrolytes, RCA seems to be a safe anticoagulation method with a favorable filter lifespan in patients underwent CRRT.
Enzo Vásquez MD
Nephrology Fellow. Department of Nephrology.
National Institute of Cardiology Mexico.


