Welcome to the 17th case of the Skeleton Key Group, a team of 40-odd nephrology fellows who work together to build a monthly education package for the Renal Fellow Network. The cases are actual cases (without patient identifying information) that intrigued the treating fellow.
Written by: Kinjel Shastri
Visual Abstract: Denisse Arellano
A. The Stem
A 71-year-old man with a history of CAD, CVA with right-sided deficits and dysphasia, neurogenic bladder with self-catheterization, and type 2 diabetes mellitus presents from a nursing home for altered mental status and hypoxia. He has a history of CLL with prominent B cell type morphology.
At baseline, he is alert and oriented, but for the past 5 days has become progressively more confused. His oxygen saturation was between 85-89% on room-air. Initial labs are found below:
Vitals:
BP: 101/44 mm Hg, Pulse: 68 bpm, Respiratory Rate: 20/ min, Temp: 97.3 F (36.3 C), SpO2: 85-89% on room air
B. The Labs
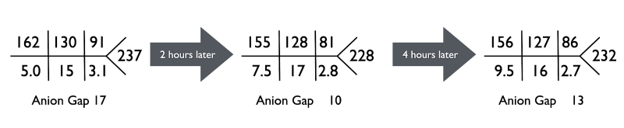
WBC (x 109/L) 4.91
Hb (g/dL) 9.1
Hct (%) 38.5
Plt (x 109/L) 119
Upon arrival to the emergency department, IV Normal Saline was started at 125 cc/hr. After the second set of labs reported a K of 7.5, Nephrology was called. Any time we get a call regarding an older man with AKI and hyperkalemia–especially given a history of neurogenic bladder–obstruction is high on the differential of reversible etiologies, so the initial reflex is to ‘place that foley (catheter)!’
Our patient had a foley placed with 350 mL of urine output, and we continued IV fluid resuscitation. The EKG showed a normal heart rate without peaked T waves, prolonged PR intervals, P wave flattening, or QRS widening. In the ED, he had already received 15 mg albuterol nebulizer, 10 units of IV insulin with 1 ampule of dextrose 50%, and 1000 mg of calcium gluconate as temporizing measures for hyperkalemia. In addition to that, we gave 80 mg IV furosemide and 0.2 mg of fludrocortisone for increased kaliuresis.
C. How do we approach hyperkalemia?
We can break it down into 3 categories (or buckets as previously described in our 12th case):
- Reduced elimination (e.g. low GFR, aldosterone deficiency, RAAS blockers, NSAIDs, potassium sparing diuretics, inherited disorders like Gordon syndrome)
- Increased intake/ tissue release (e.g., tumor lysis syndrome, rhabdomyolysis, supplemental potassium)
- Transcellular shift (insulin deficiency, DKA, hyperosmolar state, digoxin, metabolic acidosis).
The first step when evaluating for hyperkalemia is to rule out pseudohyperkalemia, wherein certain clinical conditions may lead to in vitro lysis of cellular contents, potassium leakage out of the cells, causing spuriously elevated serum potassium levels. This may be related to technique of blood drawing, hemolysis, thrombocytosis, or leukocytosis. To further understand pseudohyperkalemia we must take a closer look at the components of whole blood.
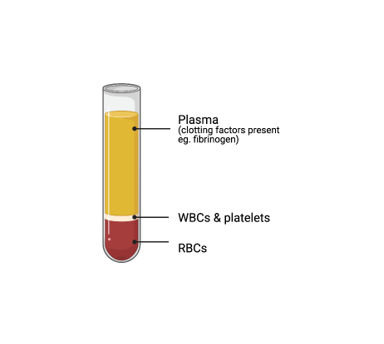
What is the difference between serum, plasma and whole blood?
We hear all these words often but let’s get into what we’re actually talking about. Whole blood refers to plasma and its clotting factors, red blood cells, white blood cells, and platelets. Plasma is the fluid component of whole blood that has clotting agents. Serum is plasma without the clotting factors: so it is made up of proteins, electrolytes, antibodies, antigens, and hormones. Serum does not require anticoagulation for separation; however, whole blood and plasma do as seen most often with heparinized tubes.
What is the difference in serum compared to blood gas analysis of electrolytes?
Electrolytes can be measured in either serum or whole blood using ion-sensing electrodes. Most often labs are reported from serum samples via auto-analyzers. In an emergency situation however, it is good to note that you can more quickly measure electrolytes in whole blood via arterial blood gas analysis. This can happen quicker in whole blood as you do not have to separate the specimen into serum as we mentioned earlier. The theory on the possible difference between serum and plasma potassium levels revolves around increased potassium release from the cells during a delay in separation of serum from whole blood, thus potentially causing a falsely elevated potassium. By eliminating the need for separation of serum from the sample, we decrease the risk of hemolysis and potassium release from the cells.
D. More Data
Baseline Cr 0.9 mg/dL
Ca: 8.2 mg/dL
Phos: 6.5 mg/dL
Mg: 2.7 mg/dL
Uric Acid: 17.5 mg/dL
CPK: 95 IUnits/L
LDH: 286 IUnits/L (reference range: 0-240 IUnits/L)
ABG: 7.31/32/58/16
Plasma potassium (arterial sample): K 3.4 mmol/L
Additionally, aside from a mildly prolonged PR interval, the EKG did not show findings we might expect with hyperkalemia, such as peaked T waves, QRS widening, or ST elevation. The CPK is within normal limits, which makes rhabdomyolysis unlikely. With a high uric acid, phosphorus, LDH and history of underlying CLL, there was a concern for tumor lysis syndrome but this also could have been attributed to cell lysis, given the fragility of the cells at this level of leukocytosis.
In the setting of hyperkalemia with serum K of 9.5, the finding of a K of 3.4 on ABG is consistent with a diagnosis of pseudohyperkalemia.
E. What is pseudohyperkalemia?
This refers to an elevation in measured serum or plasma potassium due to mechanical release of potassium from cells during phlebotomy or specimen processing. Some have defined this as a difference serum or plasma potassium of more than 0.4mmol/L. Most often it is related to poor or compromised phlebotomy technique. This can happen either during or after the blood is drawn for labs. Use of a pneumatic tubing system to transport blood specimens in hospitals can lead to hemolysis and pseudohyperkalemia. This diagnosis is confirmed by sending the sample to the lab by both the tube system and in the warm hands of a medical student.
Pseudohyperkalemia may also be seen in patients with significantly elevated white blood cell counts or severe thrombocytosis. In patients with CLL, typically when WBC count is (>120,000/microL), we often see pseudohyperkalemia secondary to increased cell fragility. Centrifugation of heparinized blood draw tubes can cause increased cell destruction with subsequent potassium release.
Potential clues to pseudohyperkalemia in this case are: profound leukocytosis and rapid changes in serum potassium without EKG changes. In fact, EKG findings in hyperkalemia are not always present; thus, a normal EKG does not totally rule it out. The CLL is important because these patients have particularly fragile WBCs that are prone to lysis.
F. Management
The patient was started on IVF 0.9NS at 125 cc/hr as he presented with hypotension and AKI. Foley was maintained, given his history of neurogenic bladder. He was started on allopurinol and the lisinopril was discontinued. The patient was diagnosed with pneumonia and treated with vancomycin and piperacillin-tazobactam. AKI was thought to be due to underlying sepsis and resolved with supportive treatment over several days.
Recognizing pseudohyperkalemia is important for two reasons: (1) it may avoid the inconvenience of starting management that may not be needed (ie. hemodialysis in this case) or (2) it may guide in prescribing the correct potassium bath in cases where maintenance dialysis is already being done.
G. Take Home Points
- Pseudohyperkalemia may be caused by an increase in cell fragility, leading to potassium leaking from the cells.
- Conditions associated with pseudohyperkalemia include leukocytosis (>100,000, specifically CLL), severe thrombocytosis (>1,000,000), prolonged tourniquet or difficult blood draw.
- Use a blood gas analyzer if there is a concern for spurious potassium measurements.

Reviewed by: Matthew A. Sparks, Joel Topf, Sudha Mannemuddhu, Chi Chu, Alejandro Meraz, Dominique Tomacruz, Dhwanil Patel, Nasim Wiegley, Ramiz Aziz, Bilal Sheikh, Jefferson Triozzi, Jamie Willows, Anna Gaddy



Very useful approach
Had a case recently. Important to note in the leukemia example, heparin can also induce lysis. This is not relevant when using blood gas analyzer has quantity of heparin in BG syringes is very small. If say a BG analyzer is not available at your hospital, using POC device at bedside can work; you have to ask for sample to be drawn carefully, not shook and collected without heparin. Thanks