 While the GI & renal systems are not typically linked together, there are a few aspects to managing a GI Bleed in patients with CKD/ESRD which are somewhat unique.
While the GI & renal systems are not typically linked together, there are a few aspects to managing a GI Bleed in patients with CKD/ESRD which are somewhat unique.
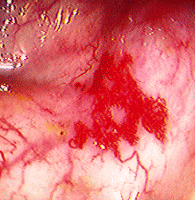
For instance, angiodysplasias in the stomach, duodenum and colon (pictured left) tend to occur at a much greater rate in ESRD patients than in the general population and should be included in the differential diagnosis. Unfortunately, angiodysplasias are notoriously difficult to locate and/or treat. Bowel ischemia, due to the increased risk of vascular disease in the ESRD population, is also a consideration.
Uremic platelet dysfunction, particularly in underdialyzed patients or advanced CKD patients who have not yet started dialysis, can contribute to GI bleeding. Strategies which can be used to improve uremic platelet dysfunction include the administration of ddAVP (given at 0.3 mcg/kg iv up to two times) or, if bleeding is expected to be more long-term, the administration of conjugated estrogens (5-30 mg iv or po daily x 5 days).
PPI’s are safe in ESRD patients, but H2-blockers (e.g., ranitidine) need to be adjusted for level of renal function. The drug sucralfate (carafate) should be avoided as it contains aluminum, and therefore if given for prolonged periods of time can accumulate in the CNS and lead to dementia. Similarly, the use of bismuth (often used in anti-H. pylori therapy) should be minimized due to its accumulation in ESRD patients.
Since many patients with ESRD are also on the transplant waiting list, it may also be prudent to transfuse these patients with leukocyte-poor pRBCs in order to minimize the risk of generating alloantibodies that might make finding a negative crossmatch that much more difficult.


Sounds challenging, and unfortunately not that uncommon.
In that AIM article I cited, angioectasias accounted for >50% of ESRD patients' GI bleeds. The article is a little dated but probably still revelant.
I have a similar ESRD pt who developed angioectasias of the small bowel and water melon stomach and inspite of multiple laser coag tx by GI still needs 2-3 units of PRBC every week. We have tried estrogens, somatostatin but nothing works.