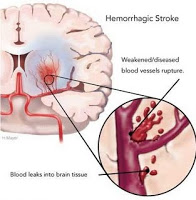
 The TREAT trial was one of the bigger stories to emerge from this years ASN. This was a large, multicenter trial of darbepoeitin (Aranesp) vs. placebo in 4000 predialysis CKD patients with type 2 diabetes and anemia. The two groups did not differ in the two primary endpoints of all-cause death or cardiovascular event and death or end-stage renal disease and on the plus side, compared with placebo, treatment with Aranesp did result in some improvement in fatigue, less need for red-cell transfusions and a reduction in cardiac revascularization. However, there was also a significantly increased risk of fatal or nonfatal stroke (5% versus 2.6%; HR 1.92, 95% CI 1.38 to 2.68), which was not explained by systolic blood pressure. The Aranesp group was treated to achieve a target hemoglobin of 13 g/dL, which is higher than I generally aim for, and it may be that a more restrictive dosing strategy could mitigate the risk of stroke. Even still, these findings create a lot of uncertainty and unease as to how best manage anemic CKD patients in the clinic. As a footnote, this study also lends fuel to the growing literature on ESA’s driving the progression of cancer. In patients with a history of malignancy at baseline, cancer death was an order of magnitude more common in the darbepoetin group (7.4% versus 0.06%, P=0.002).
The TREAT trial was one of the bigger stories to emerge from this years ASN. This was a large, multicenter trial of darbepoeitin (Aranesp) vs. placebo in 4000 predialysis CKD patients with type 2 diabetes and anemia. The two groups did not differ in the two primary endpoints of all-cause death or cardiovascular event and death or end-stage renal disease and on the plus side, compared with placebo, treatment with Aranesp did result in some improvement in fatigue, less need for red-cell transfusions and a reduction in cardiac revascularization. However, there was also a significantly increased risk of fatal or nonfatal stroke (5% versus 2.6%; HR 1.92, 95% CI 1.38 to 2.68), which was not explained by systolic blood pressure. The Aranesp group was treated to achieve a target hemoglobin of 13 g/dL, which is higher than I generally aim for, and it may be that a more restrictive dosing strategy could mitigate the risk of stroke. Even still, these findings create a lot of uncertainty and unease as to how best manage anemic CKD patients in the clinic. As a footnote, this study also lends fuel to the growing literature on ESA’s driving the progression of cancer. In patients with a history of malignancy at baseline, cancer death was an order of magnitude more common in the darbepoetin group (7.4% versus 0.06%, P=0.002).



The TREAT Trial was designed prior to the shift in treating to a target of 12 on the high end. The ESA labels have always noted a target range of 10-12.
I don't know why the recent ESA trials keep targeting a HgB of 13 or more. The FDA labeling is 10-12 and that is what the community practice is. Medicare and other carriers will pick up on this and try to restrict ESA use, even in the appropriate target range, if we are not careful. It will ultimately start restricting what may be legitimate and safe use of ESA…