You may have come across the phrase “Luminex beads” while doing your transplant nephrology rotation, or heard the term while hanging out in the Tissue Typing Lab. This post is intended to briefly describe the Luminex technology and how it works. A good review by Tait et al can be found here.
Acute cellular rejection in kidney transplant recipients comes from the presence of recipient anti-HLA antibodies directed against donor antigens. How do we detect such antibodies and prevent donor-recipient mismatches from occurring? Traditionally–and still one of the most practical tests–we use the complement-dependent cytotoxicity (CDC) “cross-match” assay. While useful, however, more recent “solid-phase” technologies have allowed a more sensitive detection of anti-HLA antibodies in recipient serum, and the Luminex system is one of them.
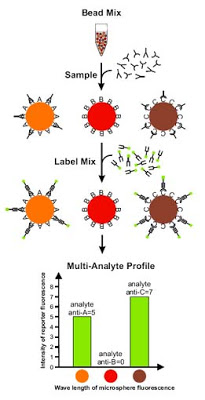
Briefly, the Luminex technology consists of a series of differently-colored polysterene beads, populations of which contain distinct HLA molecules attached to them. The lab incubates an aliquot of the kidney transplant recipient’s serum with an aliquot of beads, and if there are any anti-HLA antibodies present they will bind to the beads. A second phycoerythrin-labelled anti-human IgG antibody (aka, a “secondary antibody”) is then added, and after washing off unbound antibody, the beads are passed through a machine which works like a flow cytometer, dropping beads one-by-one through a narrow chamber. As the beads pass through the chamber, they are hit with lasers of a specific wavelength, exciting both the fluorescently-labeled secondary antibody AND the fluorochrome within the bead itself–thus allowing the detection and identification of specific recipient antibodies.
There are different types of beads: for example, some beads are coated with many different Class I or Class II molecules, designed to detect a wide range of recipient antibodies. Some beads are designed to be like a single cell, containing HLA antigens encoded by two different alleles from each of the Class I (HLA-A, B, & C) and Class II (HLA-DP, DQ, DR) loci. Finally, there are the “single antigen beads” (SAG), a bead coated with one sole antigen. This can be particularly useful in patients with a high PRA (panel reactive antibody)–you can actually identify the specific HLA antigens to which the individual is sensitized.
While potentially quite useful, it is important to also realize the limitations of this technology. While much more sensitive than the standard CDC cross-match, it is not always clear what a positive antibody result means–though in most instances, the CDC cross-match is still the primary determinant as to whether a given kidney donor-recipient pairing is deemed acceptable.


My understanding is that antibody mediated rejection is called humoral rejection and is different than cellular rejection. One is not exclusive of the other.