Cardiac and renal involvement usually manifest after the first or second decades of life. Fabry’s nephropathy initially presents as proteinuria or isosthenuria, later progressing to renal insufficiency. By age 35 years, half of male Fabry’s patients have proteinuria, and by age 50, about half have developed ESRD. While some female heterozygotes develop renal insufficiency, they do so less frequently, and rarely progress to end-stage disease.
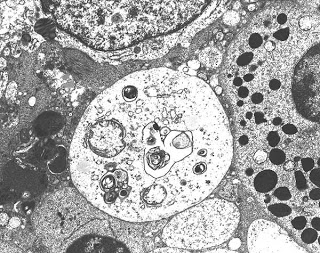
Biopsy findings in Fabry disease show lysosomal inclusion bodies containing glycolipid material in visceral epithelial cells, most prominently podocytes. “Zebra bodies”, inclusions of ceramide material in lysosomes, are often seen in the podocytes. Vascular sclerosis along with other chronic changes such as interstitial fibrosis and tubular atrophy are also commonly seen. Fabry’s can be diagnosed definitively by various serum tests measuring the alpha-galactosidase A activity. Sequencing of the GLA gene, mutations of which are known to cause Fabry’s disease, can be helpful in assessing female family members for presence of a carrier state.
Currently, two formulations of recombinant purified alpha-galactosidase A exist, although only one (Fabrazyme, alpha-galactosidase A alpha) is on the market in the U.S. While trials have not been effective in reversing renal damage in ESRD patients, they have shown a significant slowing in the rate of EGFR decrease in patients with mild renal dysfunction (EGFR > 55 mL/min). Despite the inability to rescue kidney function in our dialysis patients, however, screening plays a vital function in establishing family inheritance patterns—and identifying the next generation of Fabry’s patients in the early stages of disease. So, in younger male patients with ESRD of unclear etiology: ask about Fabry’s symptoms, screen them, and help to increase Fabry’s awareness and diagnosis!



Thank you for the comment, RFN does not give out medical advise. We suggest consultation with your medical provider.
My husband was diagnosed by the disease of Fabry and it is not capable any more of urinating, he has to carry(wear) a probe permanently for 6 months. He(it) took examinations and the doctors found nothing of abnormal in the kidney and the bladder. He(it) has problems in the heart caused by the disease. Our question is must to know if according to you why his(her,its) bladder does not work any more and if it is caused by the disease of Fabry?
Thanks to the advanceAdvance care plus
Advance relief
Helene
i have used in one patient post transplant.. the graft failed after15 yrs. dosing is adjusted per gfr..
no major interactions with transplant meds.
I read that it cost $200,000/year.There is currently a severe shortage as production has been suspended due to problems with manufacture process (contamination).(source NYT- 15TH April). Not sure if it has been used in post transplant setting.
Just curious as to the cost of Fabrazyme? Does the drug cause any problems with transplant?