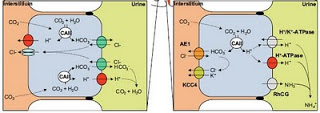
 There are two major types of intercalated cell in the collecting duct of the nephron – the alpha and beta intercalated cells. We are probably most familiar with the alpha intercalated cells in terms of acid-base handling by the kidney. They have a luminal H+ATPase enzyme and a basolateral anion exchanger. This polarity allows them to pump H+ into the lumen, facilitating acid excretion, which is equivalent to reabsorption of bicarbonate.
There are two major types of intercalated cell in the collecting duct of the nephron – the alpha and beta intercalated cells. We are probably most familiar with the alpha intercalated cells in terms of acid-base handling by the kidney. They have a luminal H+ATPase enzyme and a basolateral anion exchanger. This polarity allows them to pump H+ into the lumen, facilitating acid excretion, which is equivalent to reabsorption of bicarbonate. The beta-intercalated cells essentially have a reversed polarity – they have a protein called pendrin in their luminal membrane. This protein is a chloride-bicarbonate exchanger, facilitating chloride entry to cells in exchange for bicarbonate excretion.
As mentioned in previous posts, mutations in pendrin can cause Pendred syndrome – characterized by sensorineural deafness, hypothyroidism and goitre.
It appears that pendrin is critical for bicarbonate excretion during a metabolic alkalosis, being up-regulated during this process and down-regulated by potassium depletion (an effect that could serve as a maintenance factor in a metabolic alkalosis). Pendrin also appears to play an improtant role in chloride reabsorption in the distal tubule, with increased distal chloride delivery being associated with decreased expression of pendrin and vice-versa. Furthermore, pendrin deficient mice appear to be somewhat resistant to NaCl induced hypertension.
An interesting case report highlighted the development of a profound metabolic alkalosis in a child with Pendred syndrome, after being treated with a thiazide diuretic. Inhibition of the NaCl exchanger in the distal tubule by the thiazide would have lead to increased luminal chloride. Presumably whatever small residual amount of functioning pendrin was completely shut off by the presence of high luminal chloride. This would have limited bicarbonate excretion, facilitating development of the alkalosis.
