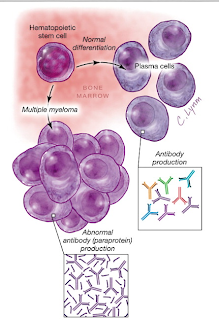
Multiple myeloma (MM) is the most common malignancy causing ESRD. Renal disease is frequently related to the characteristics of the monoclonal immunoglobulin produced, which can lead to 3 general pathological findings: “cast nephropathy’, light chain deposition disease or amyloidosis. The use of Bortezomib in combination with other agents has improved the rate of clinical remission in the past 5 years and has led to increased referrals of these patients to transplant clinics.
The challenge is that MM frequently recurs with time but patients are quite functional and strong activist of transplantation. As an example, a 55 year old female with diagnosed multiple myeloma in 2006 was evaluated for possible kidney tx. Her course was complicated by light chain deposition disease in the kidneys leading to ESRD but her MM has been on remission for the past 3 years after successful initial treatment.
Should we consider a patient with MM for kidney transplant?
Reviewing the literature, there are only few case reports and small series addressing this question. Almost all patients relapse after initial treatment if they have not undergone bone marrow transplantation. Unfortunately, many patients are not candidates for the definitive BM transplantation treatment. The topic is very controversial among different transplant centers.
Is there a difference on kidney allograft recurrence of MM depending on the type of original injury?
The graft outcomes in MM patients are determined by both the responsiveness of the underlying plasma cell disorder to initial chemo and by the inherent toxicity of the monoclonal Ig. Cast nephropathy does not seem to recur as often in the allograft compared to light chain deposition disease where the recurrence seems universal (check this review).
Is survival of patients with hx of MM improved after kidney transplantation?
No quality data available. Most of patients will have recurrence of MM (months to years after transplant) and the majority will die of either malignancy, CV disease or infectious complications. The major difficulty about doing a trial with this population is the heterogeneity of disease and low numbers of potential patients. Certain criteria to exclude high risk patients can be used to select the ideal candidate (used by Mayo but not validated): no failed prior MM therapy and absence of high risk features such as 4:14t, 14:16t, 14:20t, d17p or high risk GEP. Another prognostic marker is the evaluation of beta-2-microglobulin and albumin levels at diagnosis (high and low, respectively -> poorer prognosis). Recent European registry data reported that 1.4% of MM pts underwent renal transplantation and their mean survival was 9.4 years, while patients that remained on dialysis had mean survival of 0.91 years. Unfortunately, no information about type and severity of MM was available (inherent selection bias).
The bottom line is that kidney transplantation could benefit well-selected MM patients on remission after initial therapy and with absent high-risk prognostic markers. The final decision should take into account the risks willing to take by both patient and transplant program. Earlier recurrence of multiple myeloma with immunesuppression and increased mortality by infections are special concerns.
Credits for Dr Leung (Mayo), Dr Chandraker (BWH), Dr Heher (MGH) and Dr Scandling (Stanford) for thoughtful discussion.


You are right Kenar. Allo- BM tx would be the ideal treatment strategy, however some trials have shown too high mortality in this group compared with auto-tx, which requires less conditioning with chemo/total body irradiation. Therefore, only younger candidates and on a clinical trial setting are eligible for allo-.
MGH has been transplanting allo- kidney/BM with good outcomes. Nate had posted on this: http://renalfellow.blogspot.com/2009/04/combined-kidneybone-marrow-transplants.html. The requirement of a compatible donor is also a limitation for that.
I would think that a combined HSCT( allo) from the same donor as the Kidney Transplant would be an ideal situation? what do people think? Although, in the heme/onc world, for MM- auto HSCT is preffered.