Recently, I evaluated an older gentleman with diabetic nephropathy who had undergone a living related kidney transplant from a family member. He underwent Roux-en-Y bypass with an approximate 80 lbs. weight loss prior to his transplant to achieve a BMI of 30. His post transplant allograft function was initially stable with a serum creatinine of 1.5 mg/dl.
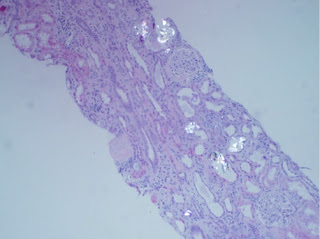
Approximately 1.5 years after his transplant he was lost to follow up and ceased his surveillance laboratory testing. He then developed worsening edema and fatigue and returned to our center for evaluation of his symptoms. On routine laboratory testing his serum creatinine was elevated at 6.8 mg/dl. He underwent an ultrasound guided transplant biopsy which revealed the findings seen in the biopsy on the left.
Pathology noted severe chronic interstitial fibrosis with evidence of oxalate deposition (shiny deposits under polarized light) within his allograft supportive of secondary oxalate nephropathy. Secondary oxalosis can occur from either high oxalate consumption (e.g. mega-doses of vitamin C, large consumption of star fruit or rhubarb) or increased enteric absorption of oxalate from either malabsorption or alterations in enteric flora (see nice editorial in the July 2012 nephsap).
following Roux-en-Y bypass was suspected as the underlying cause of the secondary
oxalosis. In a case series performed at the Mayo Clinic Roux-en-Y bypass resulted in enteric fat malabsorption
(from 4 grams per day to 9 grams per day, normal reference 2-7 grams/day) and an
increase in serum oxalate levels for 12 months, the length of the evaluation.
Urinary oxalate levels were also elevated, and enteric hyperabsorption was
shown by providing an oxalate load and demonstrating an increase in serum
oxalate levels.
timed fecal fat quantification with elevated serum and urinary oxalate levels. Unfortunately given the chronicity of renal
injury, recovery of his allograft was unlikely and he restarted maintenance
hemodialysis.
Author: Erik Lum, MD



This is such a frustrating disorder. I have started 3 patients with enteric hyperoxaluria on dialysis in the last 3 years, all in their native kidneys, all due to short bowel syndrome secondary to Crohn disease. The literature continues to ignore this disorder of bariatric surgery however, perhaps because it is interestingly not present until a year or so after surgery, suggesting that it is not simply due to fat malabsorption but perhaps some adaptation of the bowel. Recent data suggest that gastric banding does not cause hyperoxaluria as might have been expected. The optimal or effective treatment of this disorder is not really studied.