In this months edition of JCI a group led by Dr B Murphy from Icahn School of Medicine at Mount Sinai describe a beautiful set of experiments that explain the mechanism by which the CKD and eGFR risk allele rs17319721 causes chronic kidney allograft damage.
 The single nucleotide polymorphism rs17319721 is in intron 1 of a gene called SHROOM3. The risk allele A (major allele G) of rs17319721 was found in large GWAS studies of European ancestry to be associated with GFR (p=1*10−12), incident CKD (p=0.005) and GFR in type 2 diabetic patients (P= 3.18E-03). The risk allele, A, frequency is about 40% in caucasian populations but is less frequent in non-caucasian populations.
The single nucleotide polymorphism rs17319721 is in intron 1 of a gene called SHROOM3. The risk allele A (major allele G) of rs17319721 was found in large GWAS studies of European ancestry to be associated with GFR (p=1*10−12), incident CKD (p=0.005) and GFR in type 2 diabetic patients (P= 3.18E-03). The risk allele, A, frequency is about 40% in caucasian populations but is less frequent in non-caucasian populations.
The authors decided to investigate the effect of rs17319721, the SHROOM3 risk genotype, on kidney allograft fibrosis and chronic allograft nephropathy (CAN). Furthermore, they sought to determine what role, if any, SHROOM3, plays in allograft fibrosis.
The risk locus was genotyped in over 500 allograft recipients and 500 allograft donors from the GoCAR transplant cohort. Also, SHROOM3 transcript levels in 3month protocol allograft biopsies were recorded in some of these patients. Donor genotype carrying one risk allele A (A/A or A/G) was associated with higher 3month SHROOM3 expression levels compared to the normal donor G/G genotype. Interestingly, correlation occurred only in Caucasian donors when analyzed separately and there was no association with the recipient risk genotype.
The authors then looked at 12month allograft GFR and chronic allograft dysfunction index score at 12 months (CADI-12). 3month SHROOM3 expression levels were inversely related to 12 month GFR, predictive of CADI-12 and were predictive of worsening CADI score (3m to 12m)(termed ‘progressors’). These associations were not found in non-Caucasian donors.
To assess whether 3M SHROOM3 levels could predict CAN they generated logistic models that included recipient age, sex, race, AR and CIT with or without 3M SHROOM3 levels to predict 12M CADI ≥2 or 3-12M CADI change (ΔCADI) of ≥2. AUC for prediction of high CADI-12 and ΔCADI were improved in each subgroup when SHROOM3-3M level was added. For Caucasian donors AUC for ‘progression’ was 0.81 with SHROOM3 and 0.74 without SHROOM3. Furthermore, the A allele in the donor was associated with greater risk of CADI-12≥2 in all allografts (OR 1.98; CI, 1.10–3.59), indicating a higher risk of CAN with the risk allele.
This work demonstrates that the A risk allele (rs17319721) in donors is associated with higher 3M SHROOM3 levels and increase risk of CAN. Also, 3M SHROOM3 levels predict CAN and 12M GFR.
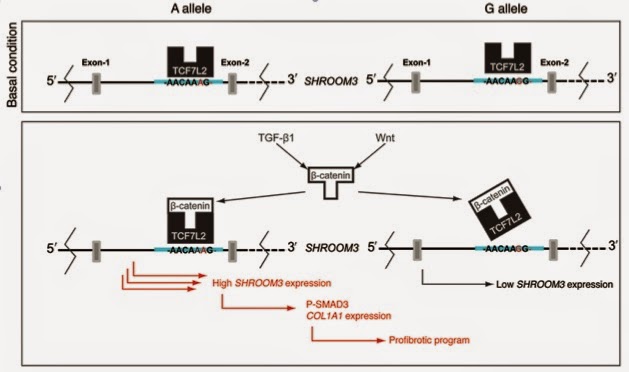
So what is the mechanistic consequence of having the A allele vs the G allele? rs17319721 is located in a transcription factor binding motif. The authors found that the transcription factor TCF7L2 binds more strongly to this motif when A is present vs when G is present. Wnt agonist increased TCF7L2/β-catenin complex binding to the A allele binding site but not the G allele site. TGF-β1 is a known key growth factor regulating renal fibrosis. The authors found that TGF-β1 induced increases in SHROOM3 expression via the Wnt/β-catenin/TGF-β1 pathway in renal tubular cells.
Then they showed SHROOM3, in turn, enhances the TGF-β1/SMAD3–induced expression of profibrotic genes including CTGF, Vimentin, and Collagen IV (downstream targets of TGF-β1/SMAD3 signaling) and these genes were significantly upregulated in allografts within the highest quartile of SHROOM3 expression.
Finally the authors verified these data in a mouse model of fibrosis.
Taken together, this data suggest an increased profibrotic program in the presence of the enhancer function of the risk allele and/or increased SHROOM3 expression. This schema is illustrated below.
This paper nicely describes the mechanism of action conferred by a single risk allele found in large GWAS studies. Until recently there has been little data to explain the relevance of the many risk SNPs described in GWAS studies. Without an understanding of the mechanism through which these SNPs confer disease there can be no progress towards identifying potential therapeutic targets. This study has identified SHROOM3 as a potential therapeutic target for chronic allograft nephropathy.