A middle-aged man with a history of microcytic anemia, well-controlled diabetes mellitus (DM)/hypertension (for 15 years), and a 16 month history of diarrhea with a 30-pound weight loss, and a creatinine of 1.9 mg/dL.
Laboratory tests showed a urine protein to creatinine ratio of 0.35 mg/g, hemoglobin of 9.3 g/dL, white blood cell count of 13.5 x 10^9/L, and a platelet count of 662 x 10^9/L. A workup for chronic diarrhea revealed an elevated erythrocyte sedimentation rate and C-reactive protein of 85 mm/hr and 18 mg/dL, respectively. Serologies for systemic lupus erythematosus, mixed connected tissue diseases, tuberculosis, hepatitides, and monocolonal gammoapthies were also unrevealing. Computed tomography of the abdomen, EGD, and colonoscopy were unrevealing – biopsy specimens of both the small and large bowel did not show evidence of inflammation. His urine sediment did not have red blood cells (RBCs) or RBC casts.
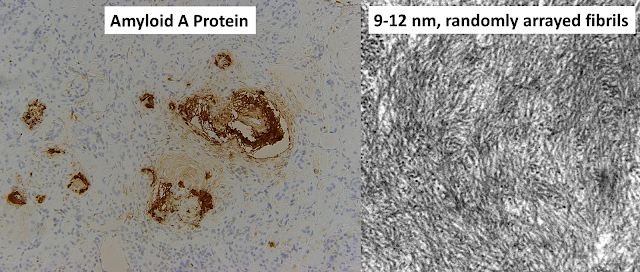
Despite his long-standing DM and hypertension, a kidney biopsy was performed – which revealed AA amyloidosis (see immunohistochemistry staining for amyloid A protein and an electron microscopy images below)
Endoscopic biopsies were subsequently stained for amyloid A protein – which was also positive.
Serum amyloid A (SAA) is part of a family of apolipoproteins associated and an acute phase reactant whose production is upregulated by the liver typically in the setting of infection or inflammation, which leads to increased expression of inflammatory cytokines like interleukin (IL)-1, IL6, and TNF-alpha. Increased SAA levels ultimately lead to the formation of amyloid fibrils that are immune to proteolysis.
The differential diagnosis of AA amyloidosis is broad – including autoimmune disorders, inflammatory arthritis, inflammatory bowel disease, lymphoma, familial Mediterranean fever, IgG4-related disease, Castleman disease, and chronic infection. A study of the natural history and outcome in systemic AA amyloidosis of a few hundred patients found that the underlying disorder was inflammatory arthritis in 60%, chronic sepsis in 15%, and periodic fever syndromes in 9%. The underlying disorder was unknown in 6% of cases.
Like most other secondary diseases processes, treatment for AA amyloidosis is treatment of the underlying disease. Other therapies should enhance clearance of amyloid deposits, decrease or interrupt fibril assembly, and decrease fibril deposition. For example, two agents that have been used with some success are a monoclonal antibody to IL-6 (tocilizumab) and the newer agent eprodisate, which directly interferes with amyloid fibril formation and deposition. SAA levels may predict disease progression and prognosis. These therapies would be potential options in this patient who did not have a proven underlying disease process.
To conclude, the presentation of amyloidosis can be nonspecific and the differential is widespread. It is important to remember that kidney biopsy can change management in a significant percentage of cases – even in a case like this, where the urinalysis was misleading and masked underlying amyloidosis.
*This is a fictionalized case based on a true account, details have been modified/changed
Samira Farouk, MD
Chief Fellow, Division of Nephrology
Icahn School of Medicine at Mt. Sinai