Lupus nephritis occurs in up to 80% of patients with SLE, and is associated with significantly reduced survival. Interestingly, one of the first studies to describe lupus nephritis was written approximately 50 years ago (Fig. 1) and was the first to utilize kidney biopsies to classify lupus nephritis based on histologic findings. This study classified cases as focal, mesangial, membranous, or diffuse glomerulonephritis, and treated the most active (diffuse) cases with high or low dose steroids. Although survival was poor in both groups (less than 2 to 5 years in most cases), this was the first study showing that patients who received steroids survived longer, and that high dose steroids showed a survival advantage over low dose.

It wasn’t until twenty years later when the 1986 NIH study evaluated steroids plus azathioprine (AZA), oral or intravenous (IV) cyclophosphamide (CYC), or a combination, for patients with active lupus nephritis. In all cases, patients also received steroids. It was clear in this study that high dose prednisone alone was inferior to any cytotoxic regimen. The study reached statistical significance for reducing the risk of ESRD for IV CYC versus prednisone alone, and this was another turning point in the treatment of lupus nephritis.
A study in 1992 compared IV methylprednisolone to short versus long courses of IV cyclophosphamide and found that again, patients who received only steroids had a higher risk of kidney failure. Interestingly, a short versus long course CYC had a similar effect on doubling of serum creatinine, but a short course of CYC was associated with a higher probability of exacerbation. A 1996 study found that that the combination of methylprednisolone and CYC was superior for remission compared to either treatment alone (Fig. 2).

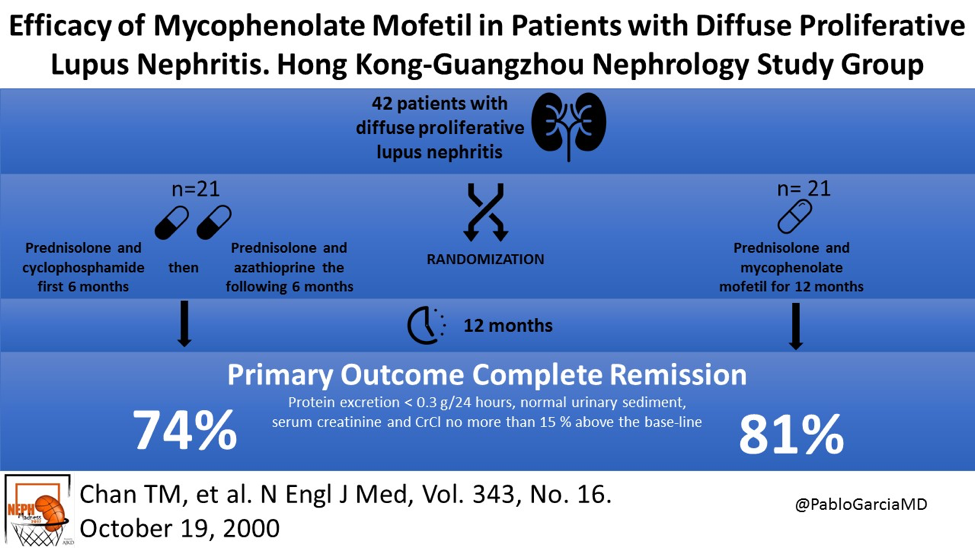
Further advances were made in 2000 when study demonstrated that induction with mycophenolate mofetil (MMF) plus prednisolone was as effective of a regimen as CYC and prednisolone followed by maintenance with AZA and prednisolone (Fig. 3). This was a truly practice-changing discovery as many patients are now treated with MMF due to a more favorable side effect profile.
The 2002 EuroLupus trial compared low or high dose CYC, in combination with steroids and AZA for all patients. Low and high dose CYC were associated with comparable outcomes.
Now, let’s talk about the ALMS trial from 2009. Continuing into an era favoring induction plus maintenance phases of treatment of lupus nephritis, several studies were published using findings from this trial. Results revealed that that MMF and IV CYC have similar effect in short-term induction, though Black and Hispanic patients responded better to MMF. A 2011 study found that showed that MMF was superior to AZA for maintenance.
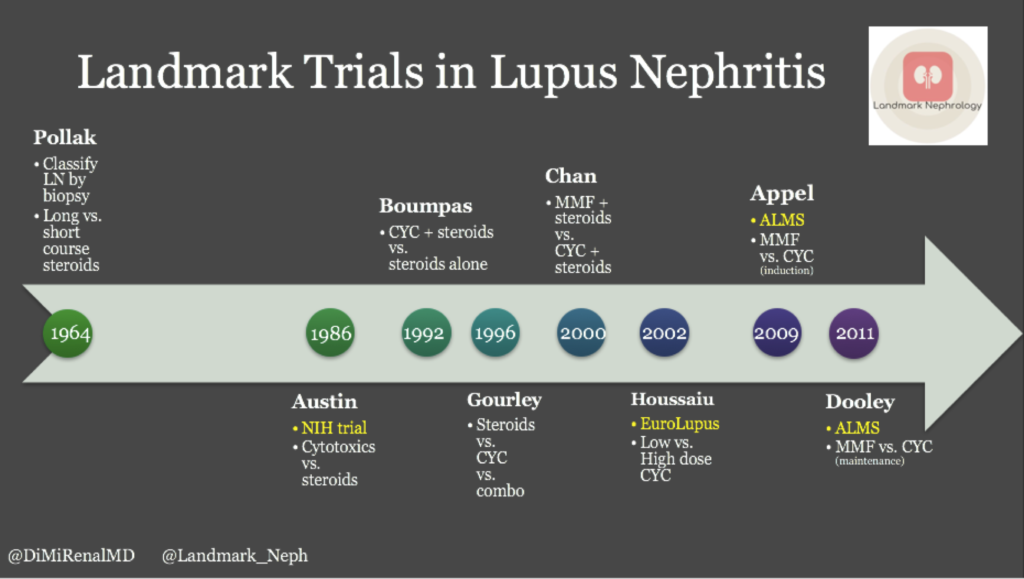
It is fascinating to look back on the history of the treatment of lupus nephritis and see how relatively recent advances in knowledge about the pathophysiology and prognosis have revolutionized treatment and increased survival (Fig 4). It can get confusing to try to compare the studies which were evaluating different doses and durations of various treatments, but an understanding of the history of treatment can prove to be beneficial for trainees and experienced nephrologists alike. Appreciation for the advances in this specific disease process brings attention to the leaps and bounds that can be made in our relatively young field and can also provide context for patients who are confused about their own disease process and their options for treatment.
Find the Spanish version of this post here.
Post by: Diana Mina, MD
Nephrologist, Dallas Renal Group
Co-Creator, Landmark Nephrology
Landmark Nephrology is an online learning tool designed to collect landmark trials in nephrology and distribute content that makes learning nephrology fun and easy.
Please visit us to check out our topic-specific content including videos, visual abstracts, quizzes, and a new slide-share portal to facilitate the exchange of educational material within the nephrology community.
We love collaboration so contact us as landmarknephrology@gmail.com or find us on Twitter to get involved!



Nice