As a nephrology trainee, everyone expects you to know all things nephron. I was having a corridor discussion with a rheumatology colleague about a recent case. He mentioned a young man who presented with acute kidney injury following cocaine use, whose kidney function was not improving. My knee jerk response was “have we checked his serum creatine kinase?” To which my colleague responds it is not that high, right around 2000 iu/L.
He then asked: What other ways can cocaine cause kidney dysfunction?
Flash back to #NephMadness2016, cocaine was a strong contender for the recreational drugs and kidney region. The ending statement for team cocaine says a lot about potential damage it can unleash: “Once the game starts…the nephrons don’t stand a chance!”
Cocaine is made from the leaves of coca plant, which grows in the Andes Mountain region of South America. Traditionally, coca leaves were chewed by ancient Incas as part of their sacred rites and rituals.
Cocaine, the alkaloid of coca leaf, was first isolated in the 1860s and became widely available in 1970s. 1-3% of adults living in developed countries have used cocaine in their lifetime. Taking into account the ever-increasing consumption of cocaine, it is important to understand the various ways cocaine can affect the kidneys.
Pharmacology
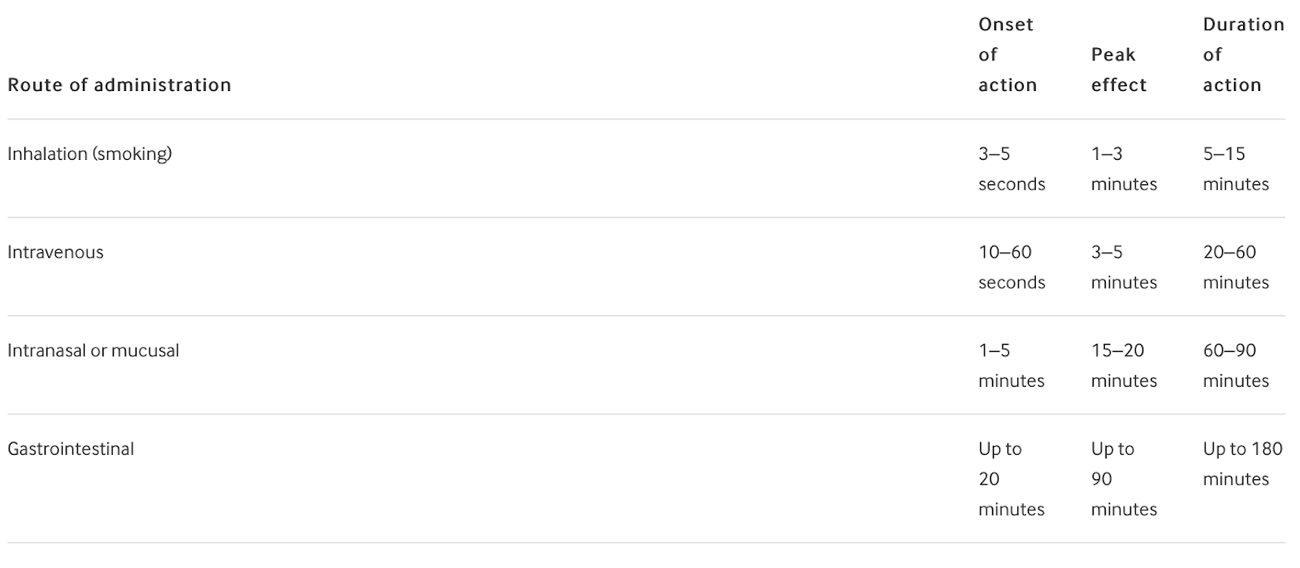
Cocaine is absorbed through mucous membranes and provides a longer duration of action when used orally or nasally compared with an intravenous route. Most ingested cocaine is metabolized by the liver with a small amount excreted unchanged in the urine after 4 to 6 hours. Metabolites are detected up to 72 hours after absorption.
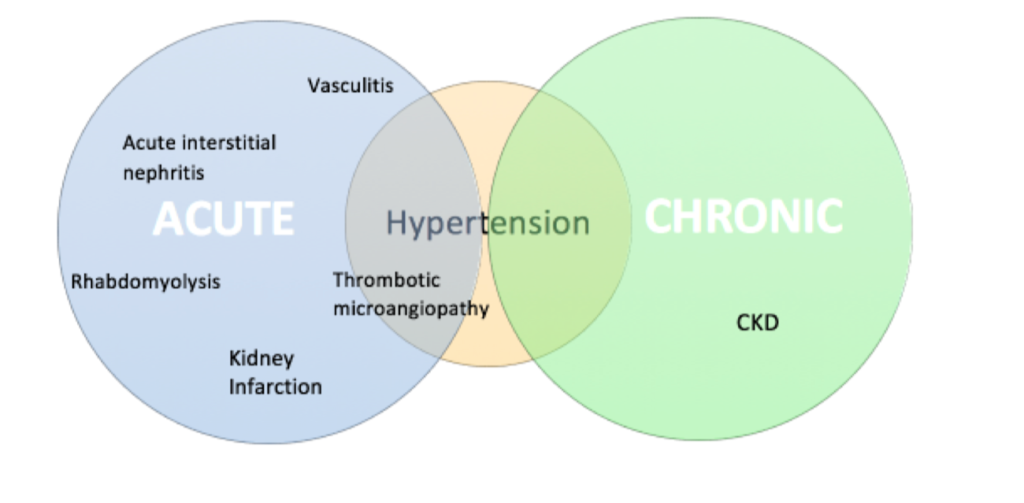
Let’s review the wide spectrum of cocaine-related kidney injury (Figure 1).
Hemodynamics
Kidney effects of cocaine are numerous and thought to be related to changes in haemodynamics, oxidative stress, and induction of renal atherogenesis. The kidneys have a rich blood supply and cocaine causes vascular smooth muscle constriction. Increased production of endothelin-1 felt to play a role. Endothelin-1 (ET-1) is secreted from vascular smooth muscle cells of renal vessels. Activation of ET-1 receptors leads to decreased renal blood flow and glomerular filtration rate. Cocaine also activates the renin angiotensin aldosterone system (RAAS). RAAS suggested because in cultured human and bovine endothelial cells, cocaine induced ET-1 release is inhibited by ace inhibitors.
Cocaine is a serotonin-norepinephrine-dopamine reuptake inhibitor, providing an additional mechanism of hypertension and tachycardia.
Rhabdomyolysis
Rhabdomyolysis is the most common cause of AKI associated with cocaine use. It is unclear why cocaine causes rhabdomyolysis. Muscle injury results from increased sympathomimetic activity induced by cocaine. Severe arterial vasoconstriction can lead to skeletal muscle ischaemia and necrosis. Increased muscle activity from seizures, hyperpyrexia and agitation secondary to cocaine consumption may also play a role in kidney injury.
Myoglobin causes renal vasoconstriction, cytotoxicity, activation of endothelin receptors tubular cell injury and tubular obstruction from heme pigment casts.
Kidney Infarction
In a few reported cases, kidney infarction has been associated with cocaine use. The underlying pathophysiology thought due to cocaines vasoconstrictive effects. Alternatively it may be due to thrombotic or embolic disease. Cocaine increases platelets aggregation and enhances thromboxane production. Other forms of vascular occlusive disease such as dissection is also described in the literature.
Vasculitis
More than half of cocaine entering the US and UK is adulterated with levamisole, an anthelmintic agent used in veterinary medicine. It was previously used as an immunomodulator in minimal change nephropathy and rheumatoid arthritis, however withdrawn due to adverse effects, notably agranulocytosis. Levamisole physically looks similar to cocaine and as a result it is often added to cut the cocaine to make it more profitable.
Levamisole increases T cell lymphocyte function and macrophage chemotaxis. It is known to induce circulating autoantibodies. Levamisole acts as a nicotinic acetylcholine receptor agonist and stimulates the sympathetic nervous system. Aminorex, the metabolite of levamisole has psychostimulatory effects similar to amphetamine.
Exposure to levamisole-adulterated cocaine can induce pauci-immune glomerulonephritis, accompanied with serological abnormalities. It presents as a clinical syndrome characterised by constitutional symptoms and cutaneous necrotising vasculitis (face commonly affected). Pulmonary haemorrhage and focal necrotizing/crescentic glomerulonephritis also reported. A wide variety of serological abnormalities have been documented; ANA, MPO, PR3. Anti double-stranded DNA, lupus anticoagulants and low complements.
Single centre data from Massachusetts General Hospital described ANCA associated disease due to contaminated cocaine use.
- All 30 patients had antibodies to MPO
- 15 had PR3 positivity
- 8 patients with abnormal urinalysis (proteinuria, haematuria, red blood cell casts)
- 3 patients with pulmonary haemorrhage (normal serum creatinine values)
- 2 patients with AKI
- 1 patient had biopsy proven pauci-immune and crescentic GN
Cocaine use was self reported in all 30 cases. Out of the 18 patients who had a urine toxicology screen, 16 were positive for cocaine and metabolites. It is not reported why urine toxicology was not performed in all patients, however this is a retrospective study and full medical records were not available in 12 cases. Authors also acknowledge lack of levamisole documentation in urine screen as a limitation. Treatment included cessation of drug and blood pressure control. Immunosuppression was initiated in the 2 cases associated with AKI. Neither required renal replacement therapy and despite immunosuppressive treatment, both had persistent CKD (eGFR <30ml/min per 1.73m2).
Other glomerulonephritis patterns have been described with cocaine use. Biopsy proven membranous nephropathy (MN) was seen with concurrent ANCA-associated vasculitis secondary to levamisole-adulterated cocaine in a small case series. Anti-phospholipase A2 receptor staining was negative in one case and not performed in the other two cases. Cocaine and levamisole were detected in the urine. Other investigations such as virology, abdominal ultrasound and chest radiography were conducted to exclude secondary MN. Treatment involved immunosuppression and blood pressure control.
Anti-glomerular basement membrane disease is postulated to be associated with cocaine use in 3 case reports. In one case, the patient presented with Goodpasture syndrome and a history of cocaine use. Anti-GBM antibodies were positive with negative MPO and PR3 antibodies. Information on urine toxicology was not given. Kidney biopsy confirmed anti-GBM disease and the patient was provided with standard therapy involving plasma exchange and immunosuppression.
Acute Interstitial Nephritis
Acute Interstitial Nephritis (AIN) is possibly an under recognized cause of acute kidney injury in patients with cocaine use. The exact mechanism is unclear but as in AIN, cessation of causative agent is key. Four case reports have described biopsy proven AIN with cocaine use. Abdominal pain, haematuria and mild proteinuria were clinical features. In all cases, kidney function recovered within a few weeks. Steroids were used in half of cases and 75% required acute renal replacement therapy.
Hypertension and CKD
Accelerated hypertension is described in cocaine use. Rarely, thrombotic microangiopathy in the setting of malignant hypertension occurs. Whether it is implicated in chronic kidney disease and chronic hypertension is less clear. Recall cocaine sets into motion haemodynamic and oxidative stress ultimately stimulating the renin-angiotensin system and causing vasoconstriction. Cocaine promotes renal arteriosclerosis. Renal arteriosclerosis is associated with end-stage renal disease.
Histological comparison of 40 kidney autopsy specimens from cocaine users were made with matched non-cocaine users. There was a significant increase in the ratio of hyaline glomeruli to normal glomerular in cocaine users compared with non-users (0.09 +/- 0.13 and 0.005 +/- 0.001 respectively).
A large epidemiological study showed no association with illicit drug use (including cocaine use) and CKD in the United states. Interestingly, a higher prevalence of hypertension was seen with cocaine use.
Another study suggests cocaine may be an important risk factor for CKD. Participants with a history of cocaine use had a greater adjusted odds of albuminuria (OR 1.80, 95% CI 1.29–2.51), but the relationship between cocaine use and reduced GFR was not statistically significant (OR 1.40, 95% CI 0.87–2.24). Lifetime cocaine use was not associated with rapid kidney function decline (OR 0.93, 95% CI 0.60–1.45). Rapid kidney function decline was defined as reduction in eGFR >3 ml/min/1.73 m2 per year over a median of 4.7 years of follow-up.
The recent CRIC (Chronic Renal Insufficiency Cohort) study showed hard illicit drug use (cocaine, heroin or methamphetamine) was associated with a higher risk of CKD progression and all-cause mortality, however it was not statistically significant for cocaine use (HR 1.28 95% CI 0.90-1.83).
Overall, the data are conflicting – making an association of cocaine use and CKD unclear with current literature.
Kidney Transplantation
Active substance use including cocaine can exclude transplantation. Substance use is a risk for medication non-adherence. Potential recipients that are actively using cocaine may need to show a period of abstinence prior to being eligible for a transplant.
Fetal Risk
Lastly, in utero. Pregnant mothers who abuse cocaine may have adverse effects on fetal kidney. Structural abnormalities on the urinary system and decrease in fetal arterial flow are observed.
Conclusion
Back to the question, what other ways can cocaine cause renal dysfunction? Yes, and in a variety of pathophysiological mechanisms I had not considered. Being aware of such spectrum of kidney injury is important given the rise in cocaine consumption particularly its adulterated form. Well-designed epidemiological studies in the future may help illuminate the relationship between cocaine and chronic kidney disease.
Post by: Renua Aba, MD
NSMC Intern, 2019



Working in Maui. two young people, same week with renal artery infarcts. Fever, flank pain, Non contrast CT negative came back and contrast study showed wedge shaped infarcts. Both using Cocaine
Excellent review on cocaine associated nephropathy. Cocaine used subcutaneously has also been found to be associated with amyloidosis