Introduction
Home hemodialysis has been around since the mid-1960s, and a study published in the Annals of Internal Medicine in 1966 by Hampers and Merrill from Boston, MA reported their 13-month experience with 4 young (age 29-44 years), male patients on home hemodialysis. They concluded that home dialysis was a “safe and practical way” of performing hemodialysis, with “long term feasibility” established with the program lasting more than a year. This was one of the earliest studies published on home hemodialysis.
Patient having dialysis at home with 5-year-old son looking on.
Since then, there have been multiple articles published demonstrating home hemodialysis to be acceptable, feasible, and well tolerated modality. Comparisons with other modalities were attempted in the initial years. This proved difficult, however, as more patients were dialyzed in-center, and finding matched cohort of patients on home dialysis was challenging. Patients on in-center hemodialysis and home hemodialysis had fundamental differences, with the home hemodialysis patients being younger, more active, and less likely to have diabetic kidney disease, and associated comorbid conditions.
With interest in home hemodialysis being renewed in the last two decades, and a push being made for wider use of the modality, more studies have been published looking at potential advantages and disadvantages of home hemodialysis compared to conventional in-center hemodialysis.
Let’s examine these studies, published regarding home hemodialysis in the last 20 years, under the following headers.
Survival and mortality
When we think of duration and dose of dialysis and mortality, the landmark trial that comes to mind is the HEMO study, which, published in 2002, showed that there was no significant mortality benefit from targeting a higher dialysis dose in those undergoing three times a week dialysis. However, studies looking at home hemodialysis raised the question of whether three times a week alone is ideal, and if there could be benefits from more frequent dialysis.
Most studies that have compared survival benefit between conventional hemodialysis, and home hemodialysis have been observational studies, bringing with them the limitations of all observational studies – the inability to account for all possible confounding factors. Despite this, these studies, summarized in the table (adapted from Weinhandl et al) below, have shown a survival benefit with home hemodialysis
| Study | Year of publication | Conventional Dialysis | Home hemodialysis | Adjustment technique | Comparison of all-cause mortality | P value |
| Woods et al | 1996 | 3 times a week (N = 3102) | Not described(N= 70) | Indirect standardization | RR: 0.56 (0.34 – 0.92)aRR: 0.58 (0.35 – 0.95)b | 0.020.03 |
| Blagg et al | 2006 | 3 times a week (N = 19) | Short Daily(N = 98) | Indirect standardization | SMR: 0.39 (0.19 – 0.51)a | <0.005 |
| Kjellstrand et al | 2008 | 3 times a week (N = 150) | Short Daily(N = 265) | Indirect standardization | RR: 0.34 (0.20 – 0.54)c | <0.001 |
| Johansen et al | 2009 | 3 times a week (N = 430) | Short Daily(N = 43) | Propensity score matching | HR: 0.64 (0.31 – 1.31)d | 0.22 |
| Johansen et al | 2009 | 3 times a week (N = 940) | Nocturnal Daily(N = 94) | Propensity score matching | HR: 0.36 (0.22 – 0.61)d | 0.0001 |
| Weinhandl et al | 2012 | 3 times a week (N = 9365) | Short Daily(N = 1873) | Propensity score matching | HR: 0.87 (0.78–0.97)e | 0.01 |
a – adjustment for age, race, sex, disease causing ESRD
b – adjustment for additional patient comorbidities (stroke, myocardial infarction, cardiac arrhythmia, peripheral vascular disease, congestive heart failure, chronic obstructive pulmonary disease, insulin therapy, and inability to walk, eat or transfer independently)
c – adjustment for age, disease causing ESRD
d – age, race, sex, primary ESRD cause, ESRD duration, BMI, number of cardiovascular comorbid conditions, Medicaid status, and cumulative admissions and days for all-cause, cardiovascular-related, and vascular access-related hospitalizations during a maximum of 12 months preceding follow-up
e – age, race, sex, BMI, primary cause of ESRD, Duration of ESRD, Diabetes, Coronary artery disease, peripheral vascular disease, other cardiovascular disease, Congestive heart failure, Cerebrovascular disease, Cancer, Dual Medicare/Medicaid eligibility, transplant waitlist registration, cumulative hospital days, cumulative EPO dose
RR: Relative risk SMR: Standardized mortality ratio HR: Hazard Ratio
The Frequent Hemodialysis Network (FHN) trial, published in 2010 and presented at ASN “Renal” Week in Denver CO, has been the only randomized control trial that attempted to compare outcomes between conventional three times a week hemodialysis, and more frequent, six times a week hemodialysis. However, it was not adequately powered to detect a difference in mortality alone, and had a composite outcome of death or increase in left ventricular mass (HR: 0.61; 95% CI: 0.46 – 0.82; P <0.001).
From the studies described above, it is evident that the data supporting a survival benefit from home hemodialysis is weak.
But dialysis isn’t just about survival, is it? Let us now look at studies that have examined other outcomes
Quality of Life
The association of hemodialysis in general with reduced quality of life has been well documented. Does home hemodialysis, or increased frequency of dialysis make a difference to the quality of life of patients?
Results from observational studies have been mixed, with some studies demonstrating an improved quality of life for patients on home hemodialysis, while others showed no difference in quality of life measures. This was summarized in a systematic review in CJASN by Suri et al in 2006.
Two randomized control trials published after this systematic review, compared quality of life measures between conventional hemodialysis and home hemodialysis.
Culleton et al published a trial in JAMA in 2007, in which 52 patients were randomized to receive nocturnal hemodialysis 6 times a week or conventional hemodialysis 3 times a week. They found that frequent nocturnal hemodialysis did not significantly improve quality of life, measured as EuroQol 5-dimension (mean difference: 0.05; 95% CI: −0.07 to 0.17; P 0.43).
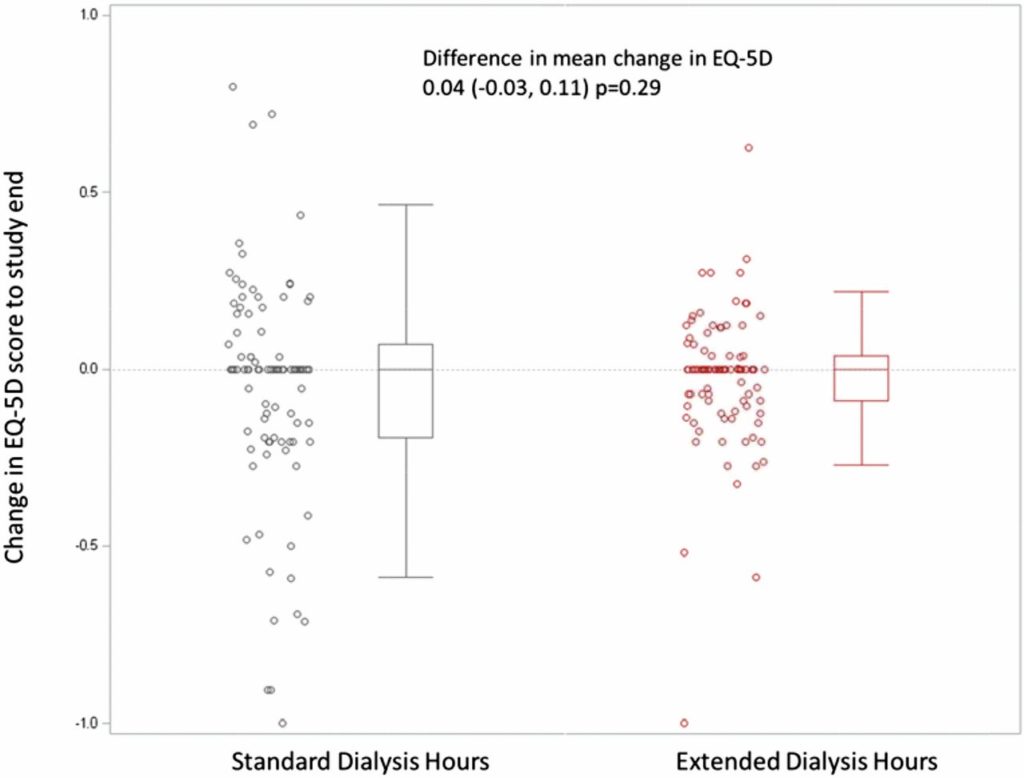
The ACTIVE trial, published in JASN in 2017 randomized 200 patients to extended weekly (≥24 hours) or standard (target 12–15 hours, maximum 18 hours) hemodialysis hours for 12 months. It found no difference in the EuroQol 5-dimension (EQ-5D) measure of quality of life between the two groups (mean difference: 0.04; 95% CI: 20.03 to 0.11; P 0.29) [This trial was covered in NephJC here].
Cardiovascular outcomes
Cardiovascular disease has a close relation with kidney disease, with cardiovascular disease being the leading cause of mortality in patients on dialysis.
What effect does home hemodialysis have on markers of cardiovascular disease?
The markers that have been most frequently studied are left ventricular mass, and blood pressure.
As is the recurring theme with data on home hemodialysis, the results on the impact of the modality on these markers are also mixed.
The three randomized control trials that have looked at the outcome of change in left ventricular mass between conventional hemodialysis and home hemodialysis.
The Culleton et al study looked at change in left ventricular mass as a primary outcome, and found a significant difference between the two groups (mean left ventricular mass difference: 15.3 g; 95% CI: 1.0 to 29.6 g; P 0.04).
As you will recall from earlier, the FHN trial looked at change in left ventricular mass as part of its composite primary outcome (along with mortality), which was reported as significantly improved in those with six times a week dialysis, and also found a significant decrease in the outcome of left ventricular mass alone in those undergoing frequent hemodialysis (adjusted mean (±SE) left ventricular mass decreased by 16.4±2.9 g in patients in the frequent-hemodialysis group, as compared with 2.6±3.2 g in patients in the conventional-hemodialysis group; P <0.001)
The ACTIVE trial, which looked at quality of life as its primary outcome, also looked at change in left ventricular mass index as a secondary outcome, and found no significant difference between the two groups (mean change at 12 months, – 16.018; 95% CI: -14.78 to 2.74 g/m2; P 0.18)
A meta-analysis by Susantitaphong et al in AJKD in 2012 looked at 38 single-arm studies, 5 crossover trials, and 3 randomized controlled trials, for the effect of home hemodialysis on cardiovascular parameters and noted a significant reduction in left ventricular mass index in the home hemodialysis groups (mean difference: 31.2 g/m2; 95% CI: 39.8 to 22.5; P 0.001).
The above meta-analysis also looked at the effect of home hemodialysis on blood pressure control, and noted a significant decrease in SBP (mean difference – 14.1 mm Hg; 95% CI: – 17.2 to – 11.0; P 0.001), a significant decrease in DBP ( – 7.1 mm Hg; 95% CI: – 9.2 to – 4.9; P 0.001), and a significant decrease in the mean number of antihypertensive medications by an average of – 0.8 (95% CI: – 1.2 to – 0.5; P 0.001).
This section cannot be completed, however, without acknowledging that the use of left ventricular mass as a surrogate outcome for cardiovascular disease outcomes in patients with CKD has been called into question – it is unclear if changes in left ventricular mass translates to change in cardiovascular mortality.
Mineral Bone Disease
Management of mineral bone disease and phosphorus control is difficult with conventional hemodialysis given the poor clearance of phosphorus with conventional thrice weekly hemodialysis. Does home hemodialysis then, by increasing the frequency of dialysis, improve mineral bone disease markers? And what does this mean for patient outcomes?
The systematic review by Suri et al in 2006 concluded that there was no statistically significant difference in the phosphorus levels or the dose of phosphate binders.
Data from the FHN trial published in 2012 by Daugirdas et al, reported a 0.46 mg/dl decrease (95% CI: 0.13–0.78 mg/dl) in mean serum phosphorus levels, and a 1.35 g/d reduction (95% CI: 0.20–2.50 g/d) in equivalent phosphorus binder dose at month 12 in the frequent hemodialysis group compared with the conventional hemodialysis group.
The other randomized controlled trials, by Culleton et al, and the ACTIVE trial, both looked at control of phosphorus and dose of phosphate binders as secondary outcomes, and concluded that the frequent hemodialysis group had a significantly lower phosphorus dose, and phosphate binder dose.
Again, while a lower phosphate binder dose might be an important outcome for patients, it is unclear if a statistically significant lowering of serum phosphorus translates to clinically significant outcomes.
Adverse events
Before concluding this review, it is important to review possible adverse effects of home hemodialysis. Data from the FHN trial was also analyzed to look at the effect of frequent hemodialysis on vascular access, and residual renal function.
Suri et al in 2013 concluded that the risk for a first access event was higher with more frequent hemodialysis than with conventional hemodialysis (HR: 1.76; 95% CI: 1.11–2.79; P 0.017), and patients on frequent hemodialysis had significantly more arterio-venous access repairs than patients on conventional hemodialysis (HR: 1.68; 95% CI: 1.13–2.51; P 0.011).
Daugirdas et al analyzed the same data to show that urine volume had declined to zero in 52% and 67% of patients at month 4 and 12, respectively in those on frequent hemodialysis, compared to 18%, and 36% in those on conventional hemodialysis, and this was statistically significant.
Conclusion
There are a few important conclusions that can be drawn at the end of this review. There have been many observational studies, and few randomized controlled trials published examining the differences between home hemodialysis and conventional in-center hemodialysis, showing mixed results in terms of outcomes including mortality, quality of life, blood pressure control, left ventricular mass, and mineral bone disease. Thus, there isn’t a body of evidence to conclusively say that home dialysis is a better form of renal replacement therapy than either conventional in-center hemodialysis or peritoneal dialysis. However, this modality has been in use for several decades now, and is at least a comparable, feasible modality, that remains severely underutilized.
Madhuri Ramakrishnan, MD
Nephrology Fellow, Washington University School of Medicine in St. Louis


