We have not talked about lung ultrasound (LUS) after the initial post about A- and B-lines last year. Amid COVID-19 pandemic, the role of LUS as a bedside diagnostic tool is expanding. It is being used to diagnose, monitor and even triage these patients. In addition, not all dyspnea is COVID and POCUS (multi-organ, when indicated) helps to rule out other potential etiologies. With more nephrologists assuming the role of front-line providers, let’s briefly discuss the common sonographic lung pathologies with an emphasis on the findings likely to be seen in COVID patients.
In part 1 of the COVID edition, we will equip you with the basics and then in part 2, go into COVID specific nuances next week.
Technical considerations:
As previously mentioned, abdominal (curvilinear) or cardiac (phased array) probes can be used to perform LUS. Some providers prefer microconvex probe if available. Whatever probe you use, make sure the depth is optimal (usually 12-15 cm) and it is aligned perpendicular to the chest wall. However, if your purpose is to evaluate the pleural sliding or look for small subpleural consolidations, a vascular (linear) probe would offer better resolution albeit at the expense of depth. Use the lung preset if your machine has one; otherwise, abdominal or cardiac presets work fine.
Interpretation
Pleural lines
Parietal and visceral pleurae appear as a single hyperechoic pleural line just deep to intercostal muscles. This line oscillates or shimmers with respiration, which represents pleural sliding. Figure 1 demonstrates a normal pleural line obtained using a linear probe. Note the sliding and relative smooth appearance without breaks in between.

A-lines
These are hyperechoic, horizontal lines arising at regular intervals from the pleural line seen in normal aerated lung [Figure 2, left panel]. They are reverberation artefacts and can simply be considered as reflections of the pleural line.
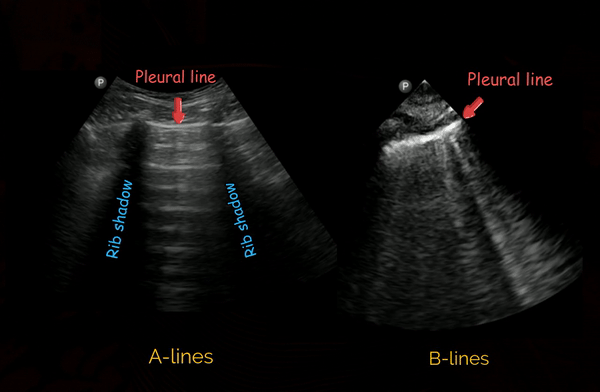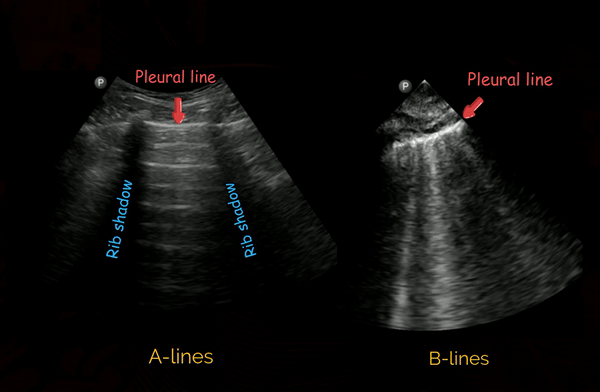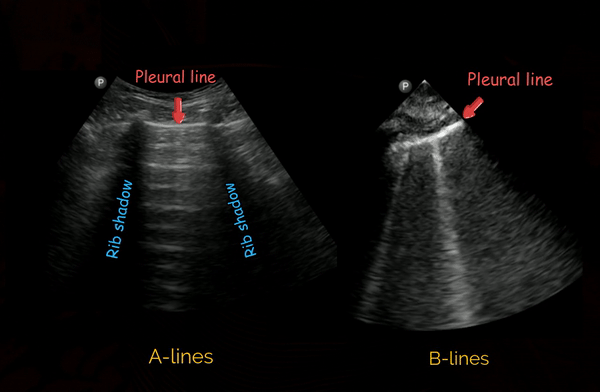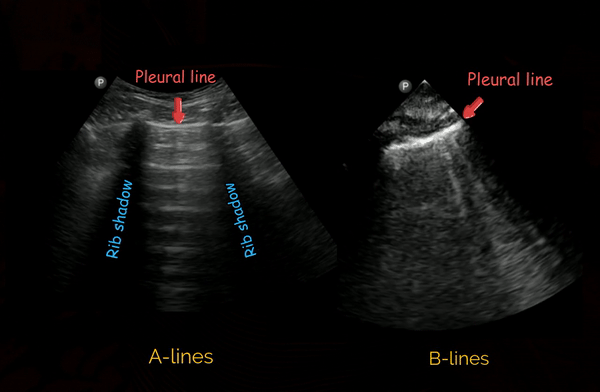
B-lines
B-lines are defined as discrete laser-like vertical hyperechoic ring-down artefacts that arise from the pleural line and extend to the bottom of the screen without fading and move synchronously with lung sliding [Figure 2, right panel]. Though the original definition says they erase A-lines, that’s not ‘always’ the case with newer ultrasound machines and the presets used. A positive B-line region constitutes presence of three or more B-lines in one rib-interspace. Two or more positive regions bilaterally is defined as ‘interstitial syndrome’ and indicates diffuse pulmonary edema, whether cardiogenic or from a pulmonary cause. In addition, B-lines are also seen in lung fibrosis and contusion. Therefore, interpretation in the right clinical context is important (consider patient’s history, B-line distribution, correlation with other POCUS findings before drawing conclusions).
Pleural effusion
LUS in fact is more sensitive than a chest radiograph for detection of pleural effusion. Non-loculated effusions accumulate in the most dependent portions of the thorax that is, the posterolateral costophrenic recesses in supine patients and anteriorly in mechanically ventilated patients in prone position. To evaluate for a pleural effusion, the probe is placed along the mid or posterior axillary line with orientation marker facing towards patient’s head and the beam is directed posteriorly [Figure 3]. Then fine sliding movements are necessary till the structures of interest are seen – liver/spleen, diaphragm, pleural fluid, lung tissue (atelectasis or pneumonia) and the chest wall. Pleural fluid appears as an anechoic area above the diaphragm surrounding the consolidated lung [Figure 4]. In addition, the thoracic spine (vertebral shadows) is often visualized above the level of the diaphragm when there is effusion, called the ‘spine sign’. Normally, the air-filled lung doesn’t allow this to happen.


Complex or exudative pleural effusion
Unlike uniformly anechoic simple effusions, complex pleural effusions often contain swirling internal echoes (known as the ‘plankton sign’) [Figure 5] and may demonstrate fibrin stranding or distinct septations (the ‘spider-web’ sign) [Figure 6]. While most complex effusions tend to be infectious in origin, a hemothorax with partially clotted blood can have similar appearance – integrating history (e.g. trauma, recent thoracentesis etc.) with POCUS findings is the key.


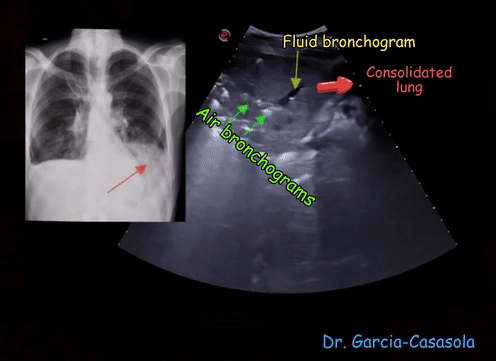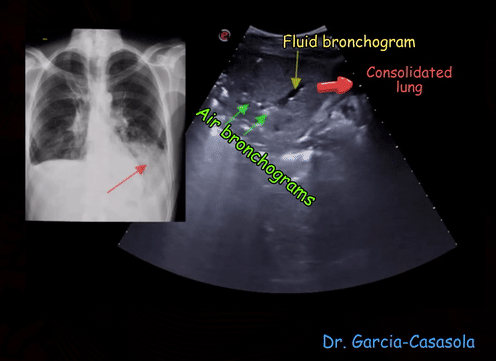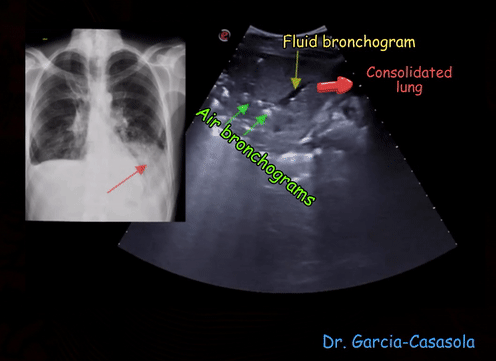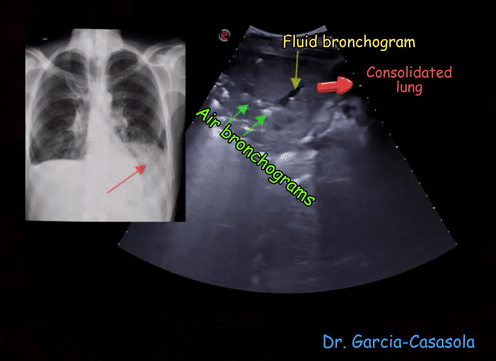
Consolidation
Consolidation often synonymously used with pneumonia, indicates solidification of the lung due to replacement of the alveolar air by exudate. In ultrasound terms, it means that the lung appears like liver in echotexture = ‘hepatization’. ‘Lobar’ or translobar consolidation is commonly seen with bacterial infection while viral infection tends to cause ‘patchy’ consolidation though this is not a dictum. White punctiform structures are often seen in consolidated lung (also in atelectasis) – these are air bronchograms (air is white on ultrasound) [Figure 7]. An air bronchogram that moves with respiration (= dynamic air bronchogram) makes pneumonia more likely than resorptive atelectasis. However, absence of dynamic air bronchograms does not exclude pneumonia and the distinction between these two entities can be difficult. In addition to the 8 scan zones mentioned in the previous post, posterior zones should be scanned when evaluating for pneumonia, particularly of viral etiology [Figure 8].

Abhilash Koratala, MD FASN (@NephroP)
Medical College of Wisconsin


