The incidence of acute brain injury, both ischemic and hemorrhagic stroke, is increasing in patients with end-stage kidney disease (ESKD). Risk factors include older age and co-morbidities requiring long-term systemic anticoagulation. Patients with acute traumatic brain injury develop acute kidney injury (AKI), with a small percentage requiring kidney replacement therapy (KRT). Performing intermittent hemodialysis in patients with acute brain injury is associated with risk of worsening cerebral edema, and nephrologists should be familiar with prescription modifications that are important to safely provide dialysis care for such patients.
Mechanism of worsening cerebral edema
Even in patients without known brain injury, intermittent hemodialysis (iHD) increases cerebral water content, leading to increased intracranial pressure (ICP). During the first hour of iHD, serum urea concentration declines rapidly in comparison to reduction in urea concentration in the brain (as time is required for urea to move across cell membranes through urea transporters). This osmolality gradient between serum and brain results in the movement of water down the concentration gradient, leading to cerebral edema. Prescription modifications could minimize changes in serum urea limiting water movement and brain edema.
The dialysate bicarbonate concentration also plays a potential role in cerebral edema and acute brain injury. During hemodialysis, as the serum bicarbonate level and arterial blood pH rise, there is paradoxical intracellular acidosis. This acidosis is the result of carbon dioxide, derived from bicarbonate, moving across cell membranes and into the cell. The intracellular acidosis leads to neuron swelling and cerebral edema, and alteration in respiratory center function.
Maintenance of cerebral perfusion
The cerebral perfusion pressure (CPP) is dependent on a stable mean arterial pressure. IHD reduces the cerebral blood flow even in stable patients undergoing routine maintenance hemodialysis treatment. Maintenance of hemodynamic stability and avoidance of intradialytic hypotension prevents further decline in cerebral blood flow. Thus, the ultrafiltration rate and total fluid volume need to be prescribed cautiously.
Timing of kidney replacement therapy initiation
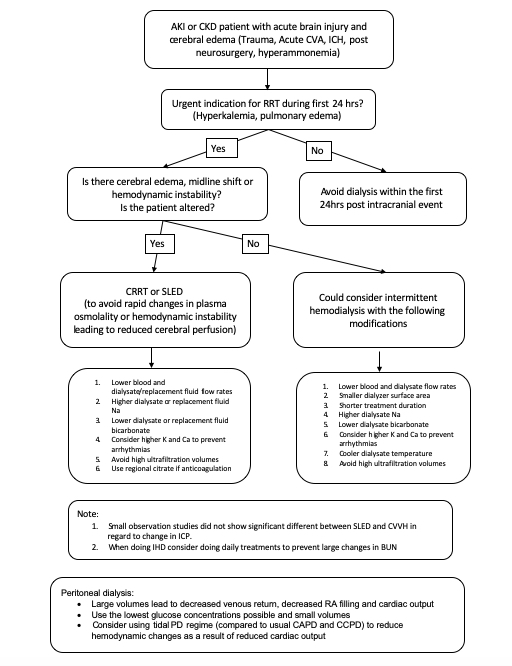
If there are no urgent indications for KRT, it may be beneficial to avoid hemodialysis during the initial 24 hours post-acute brain injury event. However, a recent observational study by Lund et al shows an association between higher initial plasma BUN level and the magnitude of ICP changes during KRT. Davenport suggests initiating KRT before a significant rise in BUN to prevent a rapid change in serum osmolality and even consider daily RRT to avoid dramatic changes in osmolality between treatments. The optimal level of pre-dialysis BUN to minimize risk of increase in ICP has not been investigated by large randomized trials, however expert opinion suggests maintaining pre-HD BUN < 30 mg/dL.
Modality of kidney replacement therapy
In patients with elevated ICP (associated with cerebral edema, midline shift, and altered level of consciousness) continuous forms of dialysis minimally change ICP and CPP, due to slower changes in plasma osmolality and hemodynamic stability (1,2,3). In a study by Ronco and colleagues, the brain imaging showed increased brain water content after IHD, whereas no such changes were observed after continuous renal replacement therapy (CRRT).
The hemodynamic and ICP effects of sustained low-efficiency dialysis (SLED) matched continuous venovenous hemofiltration (CVVH) in patients with acute intracranial hemorrhage, without the significant advantage of CVVH over SLED.
KDIGO guidelines recommend – “We suggest using CRRT, rather than intermittent RRT, for AKI patients with acute brain injury or other causes of increased intracranial pressure or generalized brain edema.” (2B)
Suggested modifications to each modality for neuroprotective measures (1,2)
CRRT or SLED
· Use of lower blood flow rate (BFR); Consider initiating treatment with lower BFR and rising gradually to 200 ml/min if hemodynamic status remains stable.
· Consider priming with albumin or other substances to reduce initial hemodynamic instability at the onset of RRT
· Avoid high ultrafiltration rates to maintain hemodynamic stability and cerebral perfusion pressure
· Higher dialysate or replacement fluid sodium concentration
· Relatively lower dialysate or replacement fluid bicarbonate concentration
· Regional citrate if anticoagulation is needed
Intermittent Hemodialysis
· IHD can be considered in patients with a minimal brain injury without significant vasogenic edema or midline shift.
· The focus is on minimizing changes in serum osmolality and maintaining cardiovascular stability.
Suggested treatment strategy for iHD:
- Combination of reduced blood and dialysate flow rates
- Smaller dialyzer surface area
- Shorter treatment session
- Use of higher dialysate sodium, calcium, and potassium for better hemodynamic and serum osmolality support
- Lower dialysate bicarbonate to avoid the rapid rise in arterial pH
- Cooler dialysate temperature for better hemodynamic support
- Avoid high ultrafiltration rates to prevent hemodynamic instability leading to reduced cerebral perfusion pressure
- Use regional citrate if anticoagulation is needed
- Consider frequent IHD treatments (such as daily IHD) to maintain serum BUN at a lower range and minimize change in serum BUN and osmolality.
Peritoneal Dialysis
Large volume exchanges of hypertonic glucose solutions can adversely impact cerebral perfusion pressure (CPP) by reducing right atrial filling and cardiac output.
· PD prescriptions:
- Use the lowest glucose concentrations possible and small volumes to avoid major swings in intraperitoneal volumes.
- For CKD patients already established on PD consider a tidal regime (compared to their usual CAPD or CCPD) to reduce hemodynamic changes as a result of reduced cardiac output.
Anticoagulation
Systemic anticoagulation with heparin or similar agents should be avoided due to the high risk of bleeding and complications. The use of citrate or other regional anticoagulants could be considered in an appropriate setting.
Sample algorithm
Nasim Wiegley, MD
Nephrology Fellow, University of California- Davis



Very enlightening this article, great work from colleagues and thanks for sharing.