I am often startled by the fact that patients are exposed to 120-200 L of dialysis solution during each dialysis treatment! Unsurprisingly then, patients on hemodialysis are particularly vulnerable to small molecular weight contaminants in the water used to prepare concentrates and dialysis fluid. These small molecular weight contaminants can enter the blood unimpeded and accumulate in the body in the absence of kidney excretion. All dialysis facilities therefore require a properly designed and maintained water treatment system to safeguard patients.
What are the potentially harmful water contaminants?
- Aluminium – added as a flocculating agent by many municipal water systems to remove nonfilterable suspended particles. It is toxic to dialysis patients, becoming sequestered in bone for long period of time, resulting in adynamic bone disease and osteomalacia. It can also cause the well-described dialysis encephalopathy syndrome.
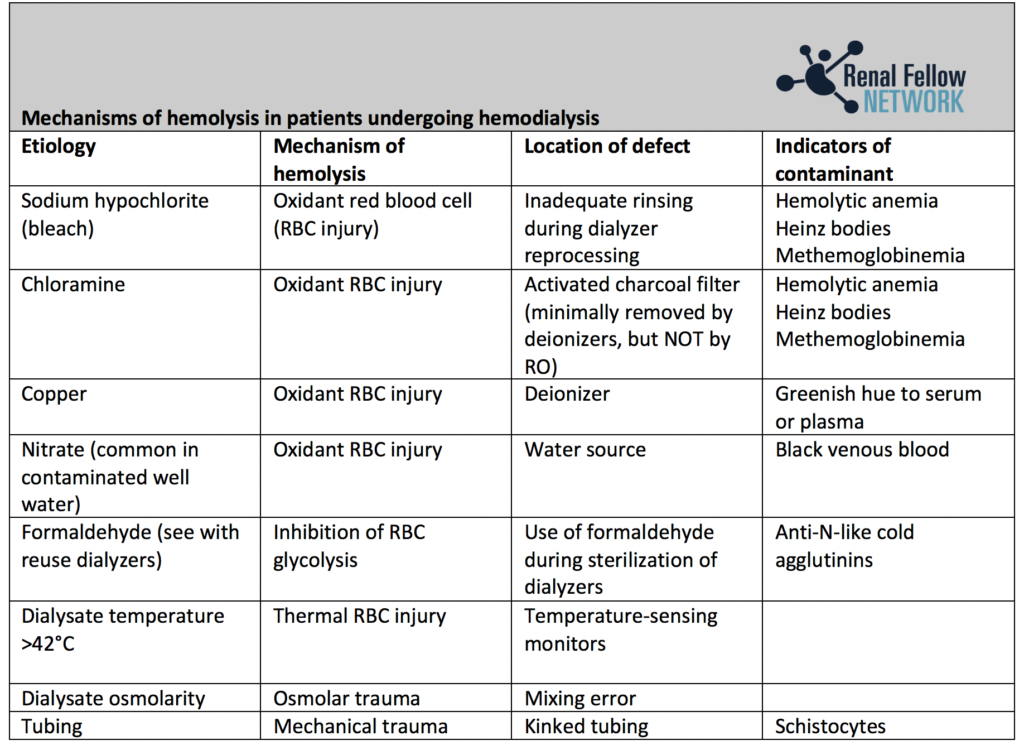
- Chloramine – added to water to prevent bacterial proliferation. It can cause hemolytic anemia (see Table 1 for other causes of hemolysis in patients on dialysis).
- Fluoride – added to water to reduce tooth decay. Large amounts of fluoride can elute from an exhausted deionizer and cause pruritis, nausea, and arrhythmias.
- Copper and zinc – can leach from metal pipes and fittings. Another cause of hemolytic anemia.
- Bacteria and endotoxin – the substances added to municipal water to suppress bacterial proliferation are removed in the water purification process for dialysis treatment. Passage of endotoxin, endotoxin fragments and other bacterial products across the dialyzer membrane and into the bloodstream can lead to pyrogenic reactions.
What are the water quality requirements?
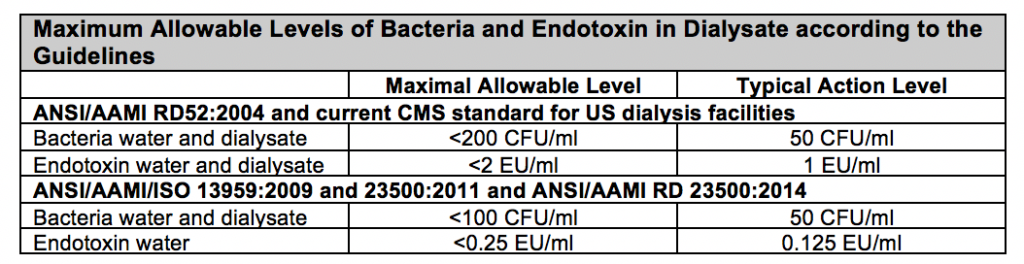
The American National Standards Institute (ANSI), Association for the Advancement of Medical Instrumentation (AAMI), and International Standards Organization (ISO) have developed minimum standards for the purity of water used to prepare dialysis solution. These are outlined the table below. The 2004 Association for the Advancement of Medical Instrumentation (AAMI) RD52 thresholds for acceptable levels of inorganic chemical contaminants in purified dialysis water have been adopted by the Centers for Medicare and Medicaid Services (CMS) in the US. However, since the original publication of RD52, the AAMI has updated its recommendations for tolerable bacterial and endotoxin concentrations in product water and dialysate. Although, the CMS continues to use the AAMI RD52 guideline to define condition for coverage compliance, this is anticipated to change and these new standards have been adopted internationally.
What is ultrapure dialysis solution?
This is characterized by a bacterial level < 0.1 CFU/ml and endotoxin level < 0.03 EU/ml. Use of very pure dialysis solution has been associated with a myriad of benefits including the reduction of the plasma levels of C-reactive protein and interleukin-6, greater responsiveness of anemia to erythopoeitin therapy, better nutrition, slower loss of residual renal function, and lower cardiovascular morbidity. It is a requirement for online convective therapies such as hemofiltration and hemodiafiltration.
What are the methods of water purification for hemodialysis?
The water treatment system is illustrated in the below figure but there are essentially three key steps:
- Pre-treatment
- Primary purification
- Distribution
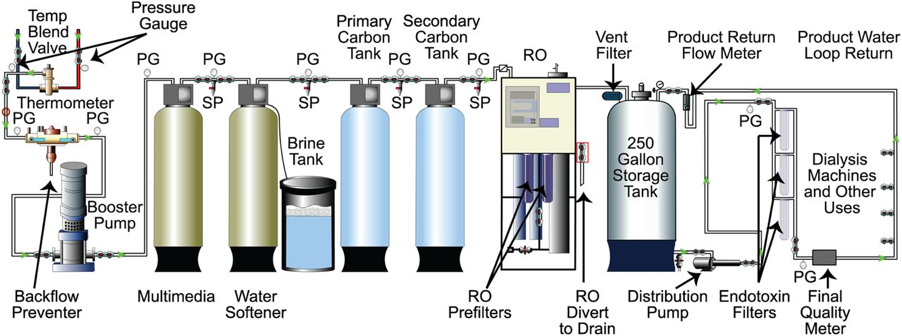
Reproduced from Ted Kasparek, and Oscar E. Rodriguez CJASN 2015;10:1061-1071
- Pre-treatment
This involves a valve to blend hot and cold water to a constant temperature, preliminary filtration, softening, and adsorption with activated carbon. Injection of hydrochloric acid to correct the pH in the case of excess alkalinity is sometimes required as this can disturb the carbon adsorption beds and the reverse osmosis (RO) membrane.
Water softeners exchange calcium and magnesium for sodium that has been affixed to a resin bed. Although these ions are also removed by RO, water softeners in regions with ‘hard water’ reduce accumulation of calcium and magnesium salts, thereby prolonging the life of the RO membrane.
Carbon adsorption by activated carbon removes chlorine, chloramines, and other dissolved organic contaminants. This step is essential and generally includes two carbon beds to ensure complete removal of chloramine
2. Primary purification process
A filter is placed just upstream to the RO membrane to catch any carbon particles and resin beads that have been inadvertently released from the pre-treatment system.
RO is the mainstay of dialysis water purification. Hydrostatic pressure drives water across a semipermeable membrane and excludes >90% of the contaminants. This strategy removes ionic contaminants, bacteria, and endotoxin.
Deionization (DI) removes ionic contaminants by exchanging cations for H+ and anions for OH–. The exchanged H+ and OH– ions then combine to become water. DI is usually used for water purification when the RO membrane fails or as an additional purification process. Water processing through DI requires bacterial control filters (ultrafilters) after DI purification because DI systems promote bacterial growth. Finally, the DI membrane is saturable and requires continuous monitoring of conductivity with mechanisms for diverting RO water when conductivity exceeds 1 microS/cm to avoid breakthrough of fatal contaminants such as fluoride.
3. Distribution of purified water
Purified water for hemodialysis is then distributed to individual dialysis machines to produce dialysate solution that remains free of contaminants. Inert material such as plastics are used to avoid chemical contaminants. Water piping systems must be carefully designed and constructed to avoid bacterial contamination with regular disinfection to prevent bacterial colonization of the system and to minimize formation of biofilm.
What monitoring is required?
There are safety standards for equipment used to purify water for dialysis as outlined by the AAMI and European Best Practices Group. Essentially, these include monitoring of the water and dialysis solution for chemical or microbiologic contaminants on a regular basis. Chloramines, for example, should be checked for at least twice daily. Dialysis patients should also be carefully monitored for any evidence of a hemolytic, pyrogenic, or other reaction, that may suggest contamination.
Post by Dearbhla Kelly



Nicely summarised
Excellent explanation
Very good
Good
Good for adequate dialysis
Very informative
Informative
Water Treatment is good prevention of water bound diseases
From my point of view as water treatment consultant, it is very understanding information and I think it is more closer to RO domestic unit.
Wishing you all the best
Emad Morsi
Good notes!
Post chlorination not required in this dialysis purification??