Focal segmental glomerulosclerosis (FSGS) is a clinicopathologic syndrome manifesting with proteinuria, usually of nephrotic range. Early in the disease process, there is a pattern of glomerular sclerosis that is focal, involving a subset of glomeruli, and segmental, involving a portion of the glomerular tuft. As the disease progresses, a more diffuse and global pattern of sclerosis evolves. Alterations of the podocyte constitute the major ultrastructural findings.
FSGS is a nonspecific histopathologic finding in several renal disorders characterized by proteinuria and progressive decline in renal function. Although it is often secondary to other disorders, including obesity, hypertension, diabetes mellitus and HIV infection, FSGS also appears as an isolated idiopathic condition. The genetic contribution to the etiology of idiopathic FSGS is indicated by reports of its occurrence in multiple members of families. Both autosomal dominant and recessive patterns of inheritance have been described.
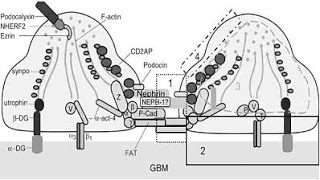
Autosomal dominant FSGS can be caused by mutations in the gene encoding alpha-actinin-4 (ACTN4) and transient receptor potential cation channel subfamily c member 6 (TRPC6). Mutations in NPHS1 (Nephrin) cause the Finnish form of congenital nephrosis and mutations in the NPHS2(Podocin) cause steroid-resistant nephrotic syndrome. Both result in a clinical picture of congenital FSGS and are transmitted in recessive fashion. Other genes implicated in glomerulosclerosis with nephrotic range proteinuria include CD2-associated protein (CD2AP), Laminin Beta-2 (Lamb2), LIM Homeobox transcription factor 1 BETA (LMX1B) and WT1.
Martin Pollak and colleagues recently reported in Nature Genetics another gene causing adult-onset FSGS with autosomal dominant inheritance. They used two large families with several affected family members in three and four generations respectively. The phenotype was defined by biopsy-proven FSGS, ESRD without cause or macro-albuminuria > 250 mg per gm creatinine. Genome wide linkage analysis defined the gene locus on chromosome 14q32. Altogether 15 genes were screened which lead to identification of non-synonymous (amino acid changing) mutations in the INF2 (Inverted Formin 2) gene in all affected family members. The investigators sequenced 91 unrelated individuals with idiopathic FSGS and identified 9 more families with mutations in INF2 all segregating with disease. Over 600 control individuals were sequenced for INF2 mutations and were negative. Most mutations were identified in the highly conserved exon 4 which encodes for DID (diaphanous inhibiting domain) of INF2.
The phenotypes in families with INF2 mutations shared certain features. Affected individuals presented in early adolescence or adulthood, typically with moderate proteinuria. Although some family members had nephrotic-range proteinuria, none of the affected individuals showed the spectrum of clinical findings that constitutes nephrotic syndrome. Microscopic hematuria and hypertension were noted in some affected individuals. Disease and proteinuria appeared to be progressive and often lead to ESRD.
INF2 is a member of the formin family of actin-regulating proteins that accelerate both polymerization and depolymerization of actin in vitro. Most mutations in FSGS were identified in DID, an autoinhibitory domain. In the most extensively studied diaphanous formin, mDia1, the interaction of the N-terminal DID domain with the C-terminal diaphanous activating domain (DAD) inhibits mDia1 function.
INF2 is highly expressed in podocytes. Electron microscopy of kidney biopsy material from an individual with INF2 mutation showed irregular podocyte foot-process morphology and prominent actin bundles. In addition, transfection studies of overexpressed wild-type and FSGS-associated mutant forms of INF2 indicate differences in the subcellular localization of the mutant and wild-type proteins, as well as differences in the pattern of distribution of actin. The precise mechanism by which actin behavior is disrupted in the presence of INF2 alterations in vivo is unclear and has yet to be defined.
Image from Podocyte Injury in Glomerular Diseases – eJIFCC 20/01 2009 http://www.ifcc.org



This is really interesting take on the concept. I never thought of it that way. I came across this site recently which I think it will be a great use of new ideas and informations.