 Another day of transplant clinic… I heard from my attending that a couple is here for a second opinion about a kidney transplant. The husband is a 60 yo male with ESRD 2/2 PKD who had a previous transplanted kidney about 10 years ago that failed last year. Due to persistent fevers, he underwent a nephrectomy of his allograft recently. On revaluation for a second transplant 1 month later, he was found to have a PRA of 98% and also positive cross-match for B and T cells against his son. His physician was not very excited about attempting another transplant and he was looking for additional recommendations.
Another day of transplant clinic… I heard from my attending that a couple is here for a second opinion about a kidney transplant. The husband is a 60 yo male with ESRD 2/2 PKD who had a previous transplanted kidney about 10 years ago that failed last year. Due to persistent fevers, he underwent a nephrectomy of his allograft recently. On revaluation for a second transplant 1 month later, he was found to have a PRA of 98% and also positive cross-match for B and T cells against his son. His physician was not very excited about attempting another transplant and he was looking for additional recommendations.
I feel that dealing with highly sensitized patients have become very common and UNOS reports that more than 25% of patients on the waiting list are sensitized and of those with PRA >80%, only 8% of patients will ever be transplanted! Today, we will discuss some of the terms used in describing sensitized patients and on my next blog will focus on the management of these patients.
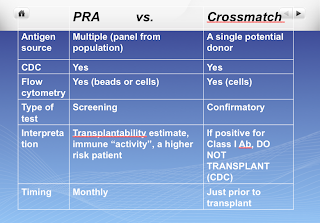
The Panel of reactive Antibody (PRA) is a screening test that can tell you how sensitized a potential transplant recipient is to HLA antigens in general. The source of HLA antigens are usually a combination of B and T cells from a panel of donors selected to represent commonly found HLA antigens in the local population. Once the serum of the recipient is added followed by complement, cell lysis detection is performed and the results are represented as the percentage of panel cells that are killed by the serum. The PRA does not tell you if you have antibody against any specific donor, but it can estimate how sensitized you might be to potential donors in your community and predict the likelihood of finding a compatible donor. For example, a PRA 70% suggests that 70% of donors will likely be unacceptable for the tested patient due to the presence of anti-HLA antibodies.
On the other hand, the crossmatch can specifically test if the recipient has any antibodies against the donor. Our patient above was very sensitized with a PRA of 98% and also had specific anti-HLA antibody against his donor. A positive crossmatch against B and T cells suggest class I anti-HLA Ab, while the presence of only Abs against B cells indicates class II. IgG antibodies against class I are the most important for transplantation. IgM antibodies usually represent autoantibodies and are not considered true sensitization. Their activity can be removed either by heating the serum to 55 degrees C or by using DTT reducing agent. If crossmatch is positive, the lab will also usually run recipients’ serum with his/her own cells to rule out auto-antibodies.
The crossmatch assays available differ in their sensitivity to detect Abs and most centers perform the classic cytotoxicity (CDC) assay, while others also perform a flow cross-match and/or luminex. In addition to determining the positivity, these assays can tell you how strong the reaction is by reporting the titer. Regardless of the test used, highly sensitized patients are often crossmatch positive to multiple potential donors and require a zero antigen mismatch allograft to increase success. These patients are therefore relegated to the deceased donor waiting list and have a very low rate of eventual transplantation.
For this reason, desensitization against preformed HLA antibodies should be considered. The titers of antibodies are important in determining how feasible a desensitization might be. We will discuss on the next blog the current recommended desensitization protocols according to anti-HLA titer levels and other potential options, focusing on our case above as an example.
Addendum: Your lab might perform additional modifications of the CDC assay: Amos-modified CDC to eliminate clinically irrelevant Abs and AHG-modified CDC, in which anti-human globulin (AHG) is added to induce cross linking and increase sensitivity.



I am a 47yo woman that has been on and off dialysis since I was 15 years old. I have had 2 kidney transplanta. 1 lasted 10 years and 1 lasted 12. The longest was a live related transplant. I am currently on dialysis for about 2 years again. I have been evaluated by my local transplant team who denied me a transplant due to my compromised vascular system- dilue to years of dialysis grafts, catheters, etc. And my PRA level is 99%. My only suggestion would be try for transplant. I plan on going to a larger transplant center to try again. The way the allocation system works now, it is possible to get a transplant with a high PRA. Worst case scenario, we will have to live our lives on dialysis. I try to just tell myself that it could be worse. I feel pretty good 4 days a week. The other days are up and down but I am blessed to be alive. Ifllts normal to feel like you cant cope with it sometimes and if you need help with it, reach out for professional help. We were dealt this and have no choice but to cope with it. I hope this helps some. You are not alone.
Hi, I saw this post and if anybody here can provide more information regarding the same. I also have high PRA (99%) and finding it hard to deal with the situation, if nybody here faced the same situation and how they coped with it?
Hi Tom, thanks for your comments. Starting with your question. The PRA is only an estimate and should not be used to draw conclusions about "transplantability". So your wife's chance of getting a transplant is not only 1%. The numbers that I showed you are from the National Organ Bank Database and are difficult to individualize to your own situation since there are multiple variables that could influence the chances of getting a transplant other than the PRA. Those include her blood type (O type is harder), availability of living donor and listing on a pair kidney exchange program. I am not sure about the availability of potential family members that could donate. If they are not matched, she could still try an organ exchange with another patient on similar situation. I would recommend you to read the following article published at the Boston Globe (http://www.boston.com/news/health/articles/2010/03/28/harvesting_hope_from_a_giving_tree/). Our goal is to get her out of dialysis as soon as possible and unfortunately staying on the deceased donor list might lead to a much longer wait. My best wishes to you and your wife! L
I am very interested in your post. My wife is 99% sensitized is only 47 y.o. and on the UNOS list and cadaver list. She just started her dialysis today. I have been trying to find out information about how the UNOS list works. I learned from her transplant coordinator that only 200 people are on the list. I don't like her odds and am planning to aggressively arming myself with information so that we can make informed decisions. Where do you get the numbers of those with PRA greater than 80% only 8% ever get transplanted. Do you have the numbers for those with PRA at 99%. Is it fair to assume that of those with PRA of 99% only 1% are ever transplanted?