 A 53-year-old Caucasian male presented with pulmonary embolism, and was found to have nephrotic syndrome. A kidney biopsy showed membranous nephropathy (MN). The disease progressed despite treatment and six years later, the patient underwent a pre-emptive living unrelated renal transplant with basiliximab induction and triple maintenance(prednisone, MMF and tacrolimus). Urine protein/creatinine ratio (U Pr/Cr) was about 1 g/g at the time of discharge. His serum creatinine (SCr) nadir at around 2 mg/dl, so a kidney biopsy was done at 1 month post-tx, showing no rejection and negative deposits on EM.
A 53-year-old Caucasian male presented with pulmonary embolism, and was found to have nephrotic syndrome. A kidney biopsy showed membranous nephropathy (MN). The disease progressed despite treatment and six years later, the patient underwent a pre-emptive living unrelated renal transplant with basiliximab induction and triple maintenance(prednisone, MMF and tacrolimus). Urine protein/creatinine ratio (U Pr/Cr) was about 1 g/g at the time of discharge. His serum creatinine (SCr) nadir at around 2 mg/dl, so a kidney biopsy was done at 1 month post-tx, showing no rejection and negative deposits on EM.
After 2 years of stable renal function and no significant proteinuria, he developed a rise in both SCr up to 3 mg/dl and U Pr/Cr up to 7 g/g. A biopsy showed recurrent membranous GN, with IF positive for granular staining with IgG1 and IgG4, and EM showing subepithelial deposits.
The two main differential diagnoses for new-onset proteinuria and renal allograft dysfunction are recurrent primary glomerulonephritis and transplant glomerulopathy (a form of chronic allograft injury which is generally believed to be immune-mediated). Idiopathic MN recurs in 30-40% of patients, usually between the 2nd and 3rd year. The outcome, as in the native kidney, can be variable, although spontaneous remission appears less common. It is tempting to speculate that transplantation selects a subtype of patients with severe disease (those who progressed to ESRD) and, therefore, patients with recurrent MN are more likely to progress than not. A study of protocol biopsies from the Mayo clinic suggested that histologic lesions of post-transplant MN persist and progress, and usually (in 86% or 12 of 14 patients) lead to clinical manifestations. The impact of recurrent MN on long-term graft survival remains unclear, and most data suggest no adverse effect, although a graft loss of 10% at ~4 years was reported in one study.
One of the exciting developments in our understanding of MN has been the identification of the antibody to M-type phospholipase A2 receptor (PLA2R), a podocyte antigen, in ~70% of patients with idiopathic MN. As has been noted previously in this blog, there is evidence suggesting that levels of the anti-PLA2R may correlate with both disease activity and the likelihood of clinical response to treatment with rituximab, a monocolonal Ab to CD20 that targets B cells. Treatment with rituximab has been shown to reduce foot process effacement and lead to regression of immune deposits in those who respond.
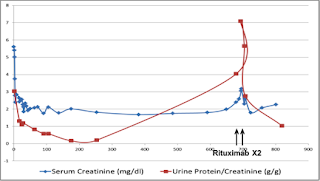
Stahl, et al reported a case of early recurrent MN in which anti-PLA2R Ab was detected pre-transplant and at the time of recurrence. Levels of the Ab fell rapidly (within 2 weeks) after treatment with rituximab, although proteinuria appeared to increase transiently (at 2 weeks), and then remit (at 2 months). It has been demonstrated in several reports that proteinuria is reduced gradually and may take several months to reach its nadir. Similarly, in our patient, we found (thanks to Dr. Becker’s lab) that anti-PLA2R, as detected by Western blot, was present in sera pre-transplant as well as time of recurrence, and disappeared completely after treatment with 2 doses of rituximab (1 g each). Both SCr (2 mg/dl) and U Pr/Cr (1 g/g) had improved by 3 months (figure above).
Some interesting questions remain unknown: 1) Are patients with MN who have detectable antibody (at the time of transplantation or later) more likely to recur? 2) How long does the response to rituximab last? (Up to 2-year data have been reported by the Mayo group) 3) Would pre-emptive treatment with rituximab of patients who have detectable antibody (at the time of transplant or later) prevent clinical recurrence, and is it worth the risk? Sindhu Chandran, MD
