 Early in 2009 Nate posted on work suggesting that renal tubular epithelial cells had the ability to repopulate the renal tubular epithelium after ischemia reperfusion injury. Since then, some amazing work has been done on what have been termed glomerular epithelial stem cells.
Early in 2009 Nate posted on work suggesting that renal tubular epithelial cells had the ability to repopulate the renal tubular epithelium after ischemia reperfusion injury. Since then, some amazing work has been done on what have been termed glomerular epithelial stem cells.
Podocytes, unlike glomerular mesangial and endothelial cells, cannot divide. However, there are a number of clinical situations such as the spontaneous remission of proteinuria in patients with membranous nephropathy and the remission of diabetic nephropathy after pancreatic transplantation that suggest that podocyte repair or perhaps even replacement via stem cells is possible.
To be termed a stem cell a cell must be capable of self renewal (the ability to go through numerous cycles of cell division while maintaining an undifferentiated state) and be able to differentiate into various specialized cell types.
Along these lines, an Italian group was able to identify candidate renal stem cells by using the cell surface markers CD133 and CD24 which are found on other adult stem cells elsewhere in the body. These cells initially were shown to be both capable of self renewal and generation of mature, functional, tubular cells with phenotypic features of proximal and distal tubules. In addition, given the right conditions these CD133 CD24 positive cells were able to become osteogenic cells, adipocytes, and cells that exhibited phenotypic and functional features of neuronal cells.
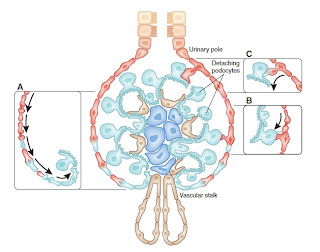
These renal stem cells are located in the urinary pole of the parietal epithelium of Bowman’s capsule. In later experiments they were shown to be able to become not only renal tubular cells but also podocytes.
In the figure on the upper left from an excellent review written by the Italian group the exquisite organization of these cells is shown. The glomerular epithelial stem cells are shown in red at the urinary pole. A transitional cell population of podocyte progenitors closer to the vascular stalk, is colored both red and light blue. These progenitors differentiate only toward the podocyte lineage and lack the properties of self-renewal. The light blue cells in the vascular stalk and glomerular tuft express only podocyte markers and the phenotypic features of differentiated podocytes.
Suggested mechanisms of podocyte replacement are also shown with A) progressive differentiation down Bowman’s capsule to the vascular stalk B) direct bridging by adjacent progenitor cells C) bridging by glomerular epithelial stem cells that subsequently acquire podocyte markers.
Excitingly, injections of these renal stem cells appear to attenuate proteinuria and histologic damage in experimental animal models of FSGS and renal dysfunction in experimental animal models of rhabdomyolysis. It’s amazing how much has transpired since Nate’s last post and hopefully, this science will help us move toward a better understanding glomerular disease and ultimately more effective treatments for our patients.

Hello,
Thanks for providing these useful tips over here. Epithelial cells help to protect or enclose organs, some produce mucus or other secretions, certain types of epithelial cells have tiny hairs called cilia, which help remove foreign substances…