Midodrine [1-(2′,5′- dimethoxy phenyl-1)-2- glycinado-ethanol (1)- hydrochloride] is a prodrug with rapid and near complete bioavailability after oral administration resulting in peak plasma levels 40 minutes after oral consumption. Midodrine is then converted by enzymatic cleavage in the systemic circulation to the active drug de-glymidodrine. This is an α-adrenergic agonist which acts by both arteriolar and venous vasoconstriction, increasing the peripheral vasculature resistance as well as venous return to the heart thereby, in theory, increasing cardiac output and preventing hypotension. On the other hand, vasoconstriction itself might also be harmful by impairing tissue perfusion, especially in the setting of pre-existing vascular disease
Midodrine has been approved by FDA to be used in orthostatic hypotension but it is also used off label for intradialytic hypotension (IDH). IDH can be troublesome in many patients, with the dialysis treatment getting disrupted multiple times, using Trendelenburg positions, turning ultrafiltration off, and not reaching dry weight. It may also result in under dialysis and is associated with higher cardiovascular events. The use of midodrine prior to dialysis sessions can improve the pre-dialysis systolic blood pressure.
In 2004, a systematic review was published on the topic by Prakash et al from London, Ontario. They selected relevant articles from Medline, Embase, ASN conference proceedings, and also obtained some unpublished data from Shire pharmaceuticals in their search for observational and clinical trials of midodrine in hemodialysis. The total number of patients was only 117 from 9 studies, with multiple confounding factors. They compared the midodrine group with the control group using fixed effects modelling and reported that the mean differences in post systolic blood pressure and diastolic blood pressures were 12.4 and 7.3 respectively. The mean differences in the nadir systolic and diastolic blood pressures were 13.3 and 5.3 respectively. All these differences were statistically significant suggesting that the use of midodrine is effective for IDH. But the authors do caution us in accepting the conclusion totally as the studies were all observational and retrospective, and studies were unblinded. Additionally, all they reported were blood pressure changes, with no clinical outcomes measured or reported
Midodrine and Blood Pressure
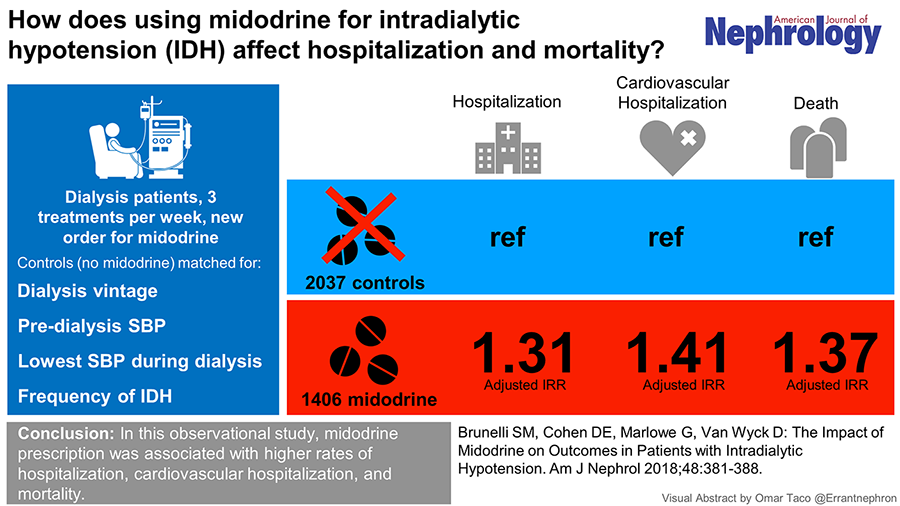
Now, we have another study which does not report changes in blood pressure, but does report clinical outcomes. Brunelli et al., studied the effect of midodrine for IDH from an administrative dataset from dialysis facilities that were participating providers with the DaVita Institute for Patient Safety, Inc.. The study included 9238 patients from DaVita clinics across United States. To compare patients who received a new midodrine prescription, they matched them each with up to 2 controls. Controls were matched according to nadir blood pressure, pre-dialysis systolic blood pressure, and mean monthly systolic blood pressure, as well as the percentage of treatments impacted by IDH. Matching was done on the basis of data in the month prior to the index date. However, they found out that as the index date approached there were differences in the groups and to eliminate these differences, they selected the cohort only from the first 10 days of the month (this resulted in reducing the midodrine group from about 3,000 to about 1,000).
Despite this, Table 1 shows that patients who received midodrine were significantly different than the matched controls who did not receive midodrine. For example, they were more likely to be older, had more Caucasians and lower albumin levels in the midodrine group, despite the matching. So what did they report in terms of outcomes? One would expect that by increasing the blood pressure and reducing intradialytic hypotension that the midodrine group would have favorable outcomes. Paradoxically, they found out that patients receiving midodrine were at a higher risk of death, all cause hospitalizations, and cardiovascular hospitalizations.
Table 1: Patient Characteristics in Unmatched, Matched, and Analytic Cohorts
Thus, after adjustment for the aforementioned covariates, midodrine use was associated with a 37% higher mortality rate compared to controls. They also reported that exposure to midodrine was associated with worse (i.e. lower) pre-dialysis blood pressure, more intradialytic hypotension episodes, similar ultrafiltration rates between the groups. Does this merely reflect residual confounding – since despite matching there were significant differences, which may persist despite an adjusted analysis with regression? The authors conclude that their findings do rule out a large clinical benefit from midodrine with respect to these outcomes. See this excellent visual abstract summarizing the study results below
Midodrine and Post-transplant outcomes
Could there be more to the midodrine story apart from these two papers? In 2016, Alhamad et al studied post kidney transplant recipients, hypothesizing that midodrine exposure in the pre-transplant period might be a risk factor for complications post-transplant. They obtained the data on 16,308 kidney transplant recipients from 2006-2008 and measured delayed graft function, graft failure, and patient death after kidney transplantation. They adjusted associations of pretransplant midodrine use with complications at 3- and 12-months post-transplant. There were 16,000 patients in the control group and 308 patients in the midodrine group. Patients who received midodrine prior to transplant were at higher risk of developing hypotension, graft failure and death posttransplant, which were statistically significant. There was also an associated higher risk of developing acute MI, ventricular arrhythmias and cardiac arrest which were also significant. A similar study done by Pottebaum et. al. showed poor graft function in patients using midodrine prior to transplant compared to nonusers as well as poor graft survival at the end of one year. None of the studies mention that midodrine is a culprit for the complications that occur but to consider midodrine use as a risk factor in predicting future complications post-transplant
Conclusion
Midodrine use appears to be associated with higher blood pressure, but with higher CV events and mortality as well. Its use may be a marker for general ‘badness’ in the association with post-transplant outcomes. Does this mean we should stop using midodrine? All these analyses are quite weak, with significant possibility of underlying selection bias and residual confounding, which cannot be completely adjusted away. After all, the sickest and most frail patients are the ones prescribed midodrine, and poor outcomes in this population should not surprise us.
Some people will call for the usual ‘more research is needed’ cliché. However, what kind of a study do we need? Obviously a properly designed randomized trial, with blinding and all. But what should be the control arm? Most importantly, what should be the outcomes? Rather than mortality, a good argument can be made for patient-important outcomes, such as quality of life, ability to stand post-dialysis, post-dialysis recovery time. These outcomes are arguably what dialysis is about.
Deepu George
Clinical Fellow in Nephrology
University of Ottawa




My nephrologist prescribed 5 mg of midodrine before dialysis. Sometimes my blood pressure is too low, so I asked him if I should increase to 10mg. He said “no” because it’s a vasoconstricter and not good for dialysis patients. I’m in the hospital and the doctor on duty has prescribed 10mg 3 times a day. I feel like this could hurt me. What do you think?