Morphological changes due to diabetes are similar in type 1 and type 2 diabetes and can be observed in all the 4 structural compartments of the kidney: glomeruli, vessels, interstitium, and tubules. Approximately one-third of diabetics will develop diabetic nephropathy.
Glomeruli:
The earliest change is diffuse thickening of the glomerular basement membranes. Diffuse increase in mesangial matrix is seen in the early phase which progresses to nodular mesangial sclerosis (Kimmelstiel-Wilson nodules) in the later stages. The nodules have a hypocellular matrix core which may be homogenous or show lamellations with peripherally arranged mesangial nuclei. The nodules are often variably sized, and are PAS and silver positive. The periphery of the nodules may show mesangiolysis resulting in the formation of capillary microaneurysms due to the detachment of anchoring points of the glomerular basement membrane to the mesangium.
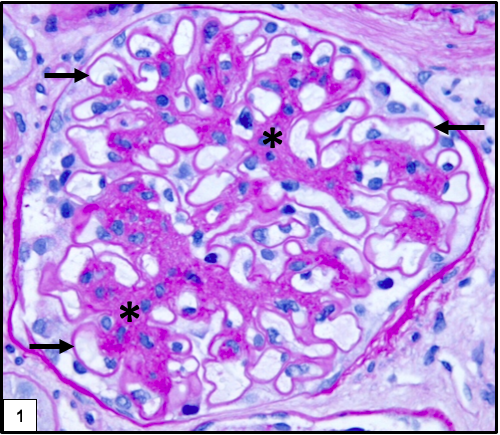
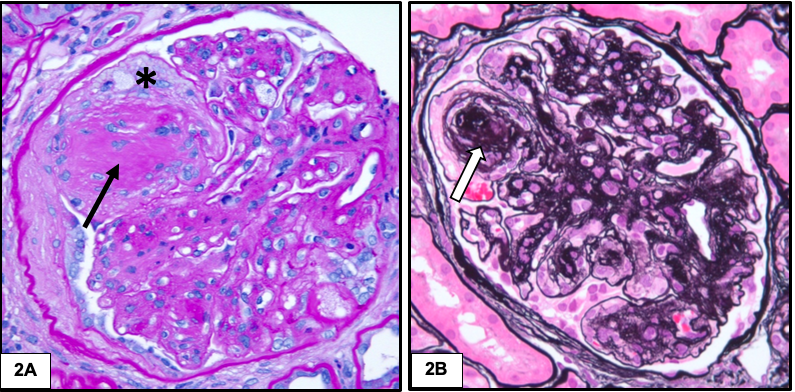
Hyalinosis lesions are common in diabetic nephropathy and result from insudation or exudation of plasma proteins from the vessels. They have a homogenous eosinophilic appearance and show two characteristic patterns: fibrin cap and capsular drop. Fibrin cap represents accumulation of plasma proteins between endothelium and glomerular basement membrane. Capsular drop is produced by accumulation of plasma proteins between parietal epithelial cells and Bowman capsules. These lesions are characteristic but not specific for diabetic nephropathy.
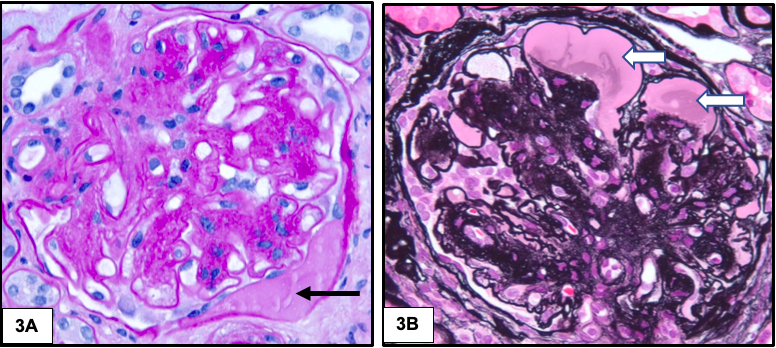
As the disease advances, segmental and global glomerular sclerosis develops.
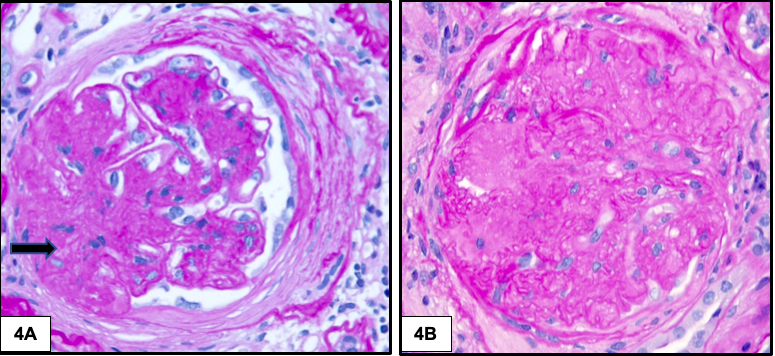
Tubules & Interstitium:
The earliest change involving the tubules is thickening of the tubular basement membranes. As the disease progresses, there is increase in the degree of interstitial fibrosis and tubular atrophy. Non-specific chronic inflammation may be seen associated with the areas of fibrosis.
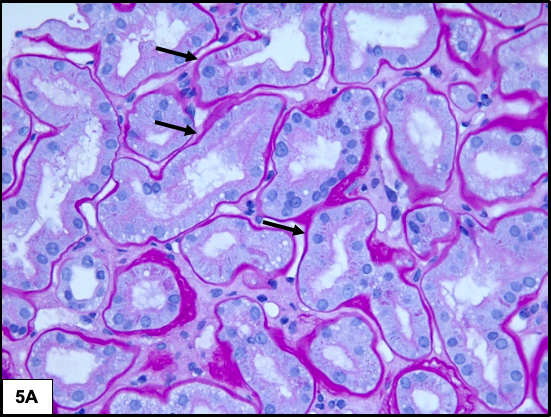
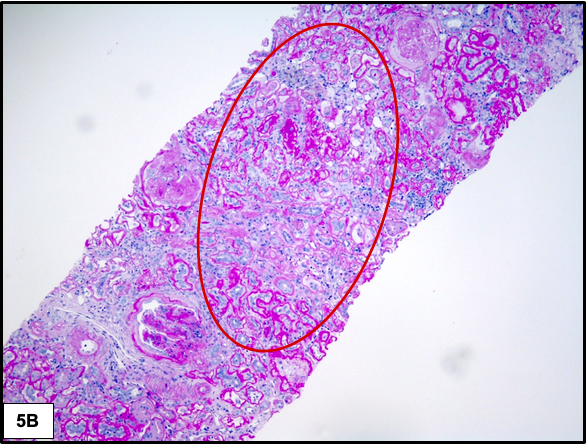
Vessels:
Arteriolar hyalinosis typically involves both the afferent and efferent arterioles and some consider this finding to be the most specific for diabetic nephropathy. Arteries show varying degrees of intimal fibrosis, but is often mild.
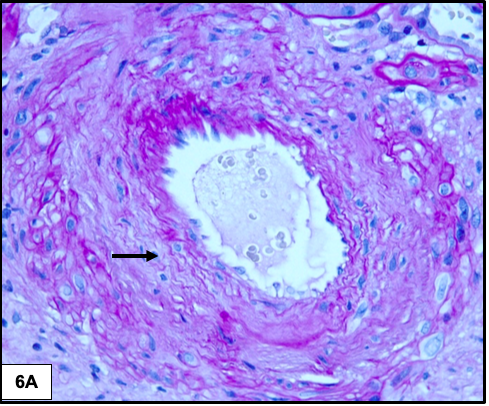
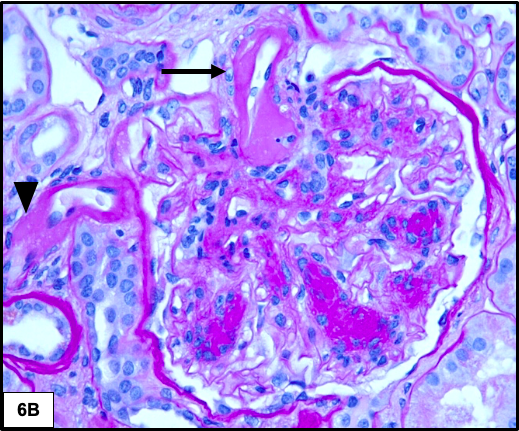
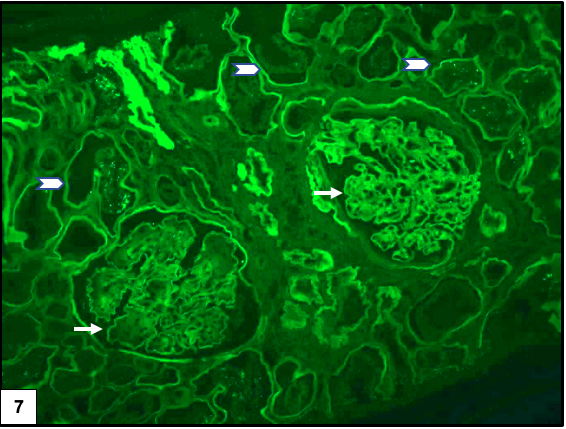
Immunofluorescence microscopy:
There is often diffuse linear staining of the capillary walls and tubular basement membranes for IgG and albumin due to non immunological trapping of proteins in the thickened, abnormal basement membranes.
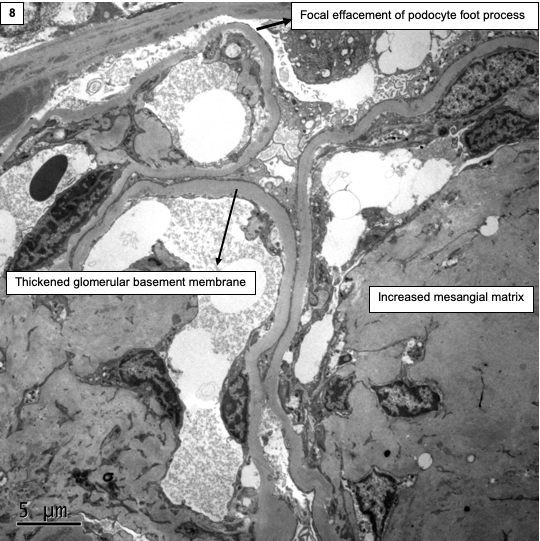
Electron microscopy:
Diffuse thickening of the glomerular and tubular basement membranes may be seen with expansion of the mesangial areas by accumulation of matrix and cellular debris. Hyalinosis lesions appear electron dense and should not be mistaken for immune-complex deposits. They are often admixed with electron lucent areas and are not as discrete as immune-complex deposits. There may be varying degree of foot process effacement.
Differential diagnosis:
Nodular mesangial sclerosis can also be seen in amyloidosis, monoclonal immunoglobulin deposition disease (MIDD), fibrillary glomerulonephritis, and idiopathic nodular glomerulosclerosis. Immunofluorescence and electron microscopy helps differentiate these conditions.
1. Amyloidosis: Deposits are Congo red positive and show characteristic non branching fibrils measuring 8-12 nm in diameter by electron microscopy.
2. Monoclonal Immunoglobulin Deposition Disease: Shows fine granular staining of the mesangium, glomerular capillary walls, and tubular basement membranes with either kappa or lambda light chains. EM shows fine granular deposits in the same locations.
3. Fibrillary glomerulonephritis: Shows granular staining of the mesangium and capillary walls with IgG and C3. EM shows non branching fibrils which are thicker than amyloid measuring 10-30 nm in diameter.
4. Idiopathic nodular glomerulosclerosis: It is a diagnosis of exclusion, there is lack of clinical history of diabetes, and it is possibly related to smoking and hypertension.
It is also important to remember that other glomerular diseases like membranous or IgA nephropathy or post-infectious GN may be superimposed on diabetic nephropathy and 45% of diabetic patients have non diabetic renal diseases.
Preethi Sekar
Renal pathology fellow, University of Chicago



Excellent. Thank you.👍
Thanks. Best histology review I have seen
Thank you so much!
Thanks for the all details about diabetic nephropathy. You have updated useful information about glomeruli, vessels, electron microscopy and the differential diagnosis.
Excellent
Thanks
Thank you very much for this illustration
Excellent review, thanks!