Welcome to the 13th case of the Skeleton Key Group, a team of twenty -odd nephrology fellows who work together to build a monthly education package for Renal Fellow Network. The cases are actual cases (without patient identifying information) that intrigued the treating fellow.
You’ve learned about the most common cause of hypercalcaemia in our 10th case from @DTomacruzMD. Now you’ve heard the horse hoofbeats, it’s time to listen out for a zebra….
Case by Jamie Willows
Visual Abstract by Corina Teodosiu
A. The Stem
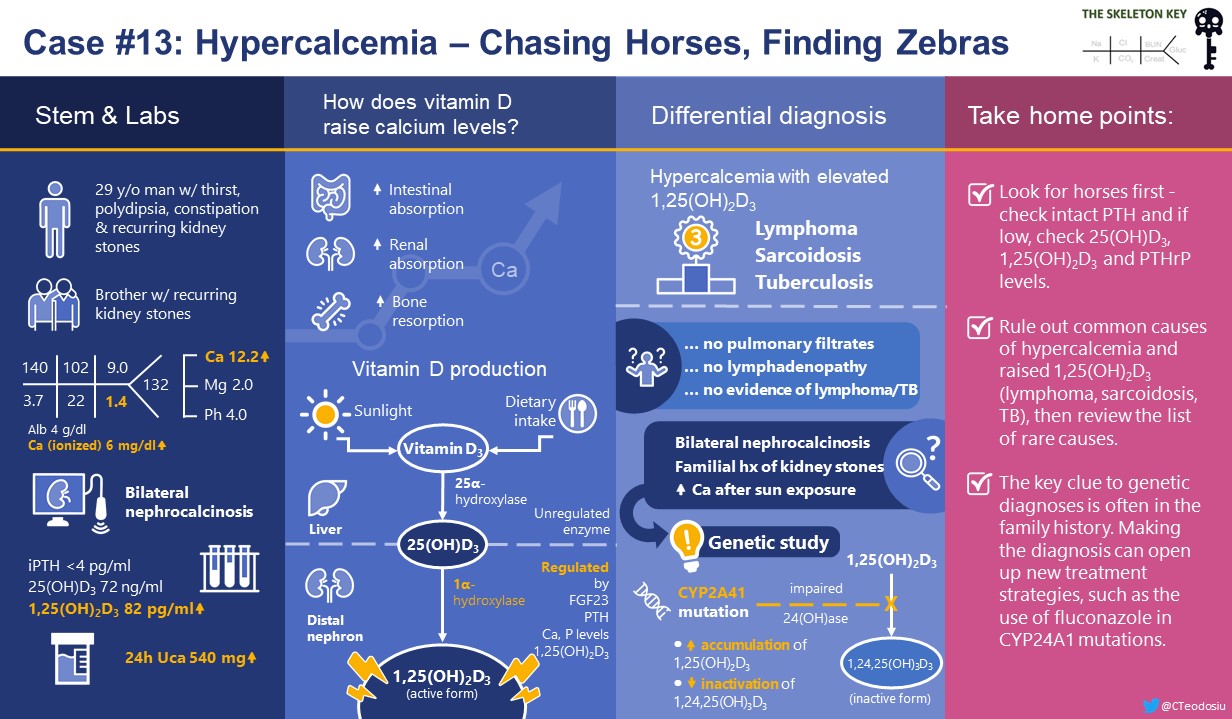
A 29-year-old man was referred to the emergency department following blood work drawn by his primary care doctor to investigate fatigue.
He reports thirst, polydipsia, and constipation for 2-3 weeks. He does not have weight loss, night sweats, cough, fever or shortness of breath. He is normally fit and well, though he had several kidney stones in his late teens which passed spontaneously. His older brother also has recurrent kidney stones. He does not take any medications or over-the-counter products, including vitamin supplements. He lives in Europe but returned the previous week from a cruise ship sailing around the Caribbean.
Vital signs:
BP 109/65 mm Hg, HR 88 bpm, RR 16/ minute, SpO2 98% without supplemental oxygen, Temp: 36.5 °C (97.7 °F)
Physical examination revealed a well-appearing white man, deeply tanned but with appropriate tan marks where he’d worn his wristwatch. Otherwise his examination, including palpation for lymphadenopathy, was unremarkable.
B. The labs
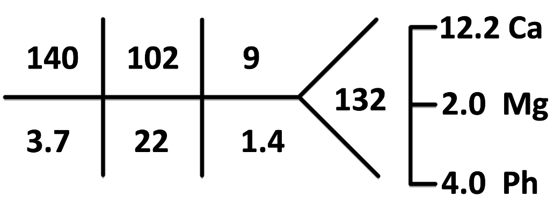
Baseline creatinine was unknown. Albumin was 4 g/dL.
Urine culture was negative.
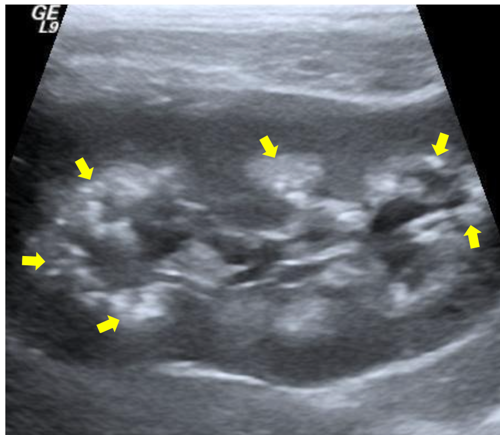
Point of care kidney ultrasound was obtained which showed medullary nephrocalcinosis bilaterally, without hydronephrosis (Figure 1).
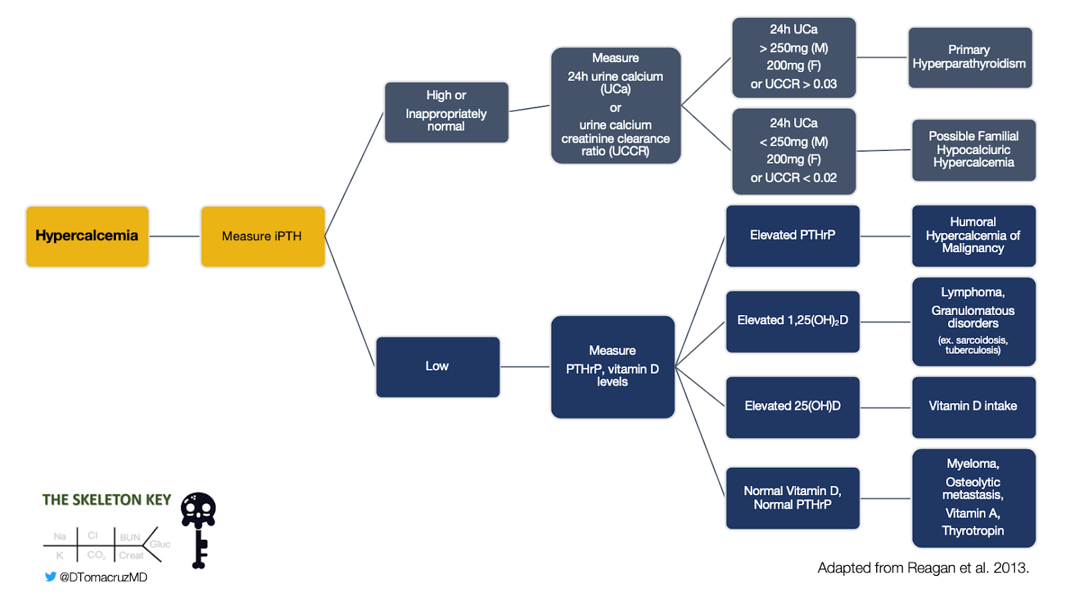
Further work-up was initiated for hypercalcemia per the algorithm from our 10th case (Figure 2), starting with measuring intact parathyroid hormone (iPTH).
He received IV fluids and was admitted to expedite investigation. Some investigations were performed prior to the parathyroid-related protein (PTHrP) and vitamin D results becoming available:
Nephrology was then consulted for further workup.
C. The Differential Diagnosis
To summarize, this 29 year old man has hypercalcamia, nephrocalcinosis and an elevated 1,25(OH)2 vitamin D level. He has just had significant sunshine exposure. He is not on medications or supplements, and has a personal and family history of kidney stones.
We have previously reviewed kidney handling of calcium under the influence of PTH, but not the effect of vitamin D on calcium homeostasis. Let’s review this first, then go back to our case.
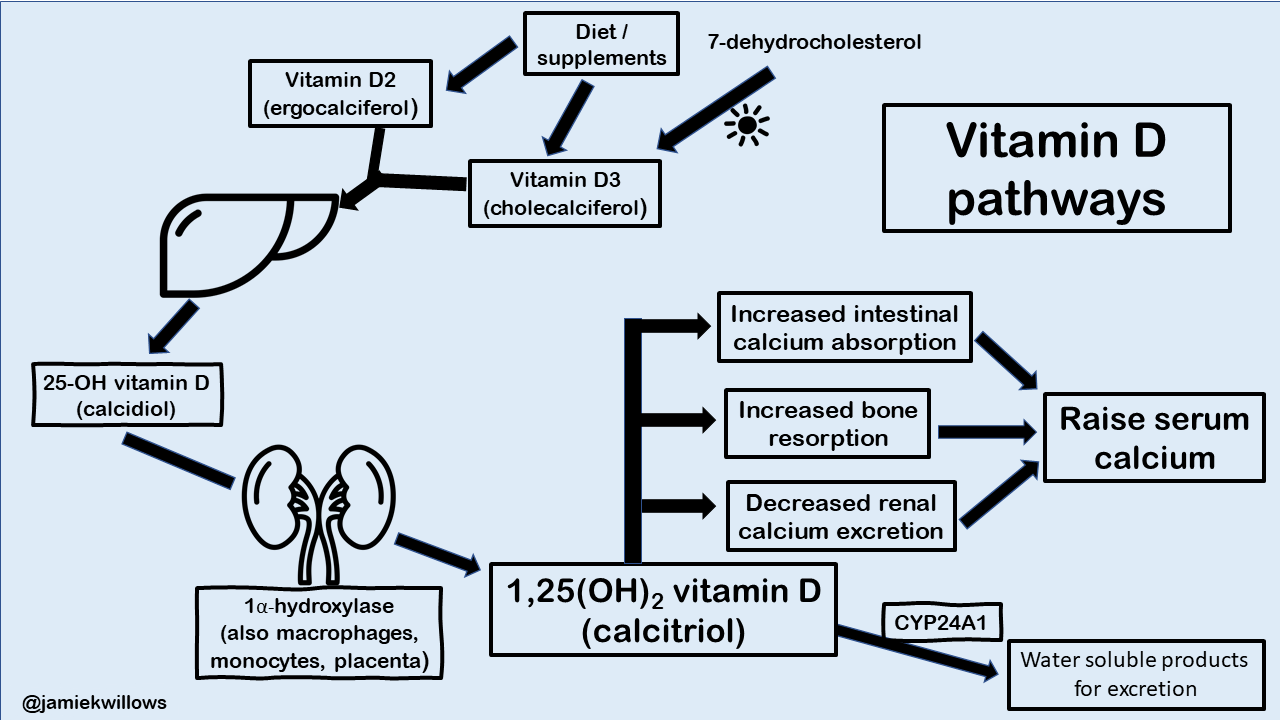
- Prior to activation, vitamin D is in the form of 25-hydroxyvitamin D, which is abbreviated to 25-OH vitamin D and is also known as calcidiol.
- 25-OH vitamin D is the form routinely measured when a serum “vitamin D level” is requested. Under usual circumstances this gives a reliable indicator of vitamin D status.
- The kidney’s distal tubules do the majority of vitamin D activation via the enzyme 1α-hydroxylase, converting 25-OH vitamin D to 1,25(OH)2 vitamin D (also known as 1,25-dihydroxyvitamin D3, or calcitriol).
- However, 1α-hydroxylase is also present in macrophages, monocytes, the placenta, skin and other tissues, so small amounts of extra-renal vitamin D activation also occurs.
- Production of 1α-hydroxylase is under tight control, primarily being stimulated by PTH.
It is 1,25(OH)2 vitamin D that is responsible for the vast majority of the biological actions of vitamin D (figure 3).
In the kidney, approximately 65% of filtered calcium is reabsorbed in the proximal tubules via vitamin D independent pathways. However, in the distal convoluted tubules, calcium reabsorption is increased by the action of 1,25(OH)2 vitamin D via the vitamin D receptor (Figure 4).
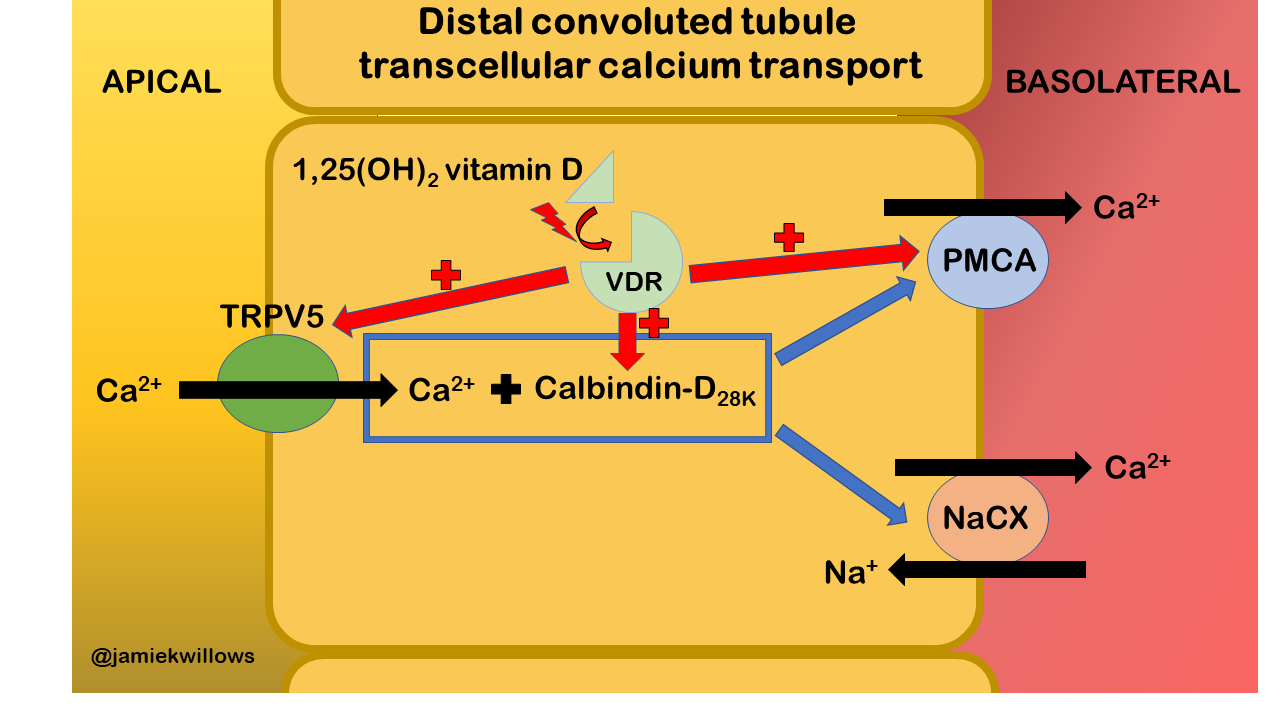
Figure 4. Effect of active vitamin D on calcium reabsorption in the distal convoluted tubule of the kidney. The TRPV5 channel is the gatekeeper for calcium entry across the apical membrane. Intracellular calcium binding proteins (including calbindin-D28K) facilitate movement of calcium across the cell. The basolateral plasma membrane calcium (PMCA) pump and the sodium-calcium exchanger (NaCX) then extrude calcium across the basolateral membrane. When the vitamin D receptor (VDR) is activated by 1,25(OH)2 vitamin D, it acts to enhance expression of PMCA, Calbindin-D28K, and to a lesser extent TRPV5. Diagram simplified from the works of Bikle, Kumar, and Christakos.
The effects of 1,25(OH)2 vitamin D on the intestine, kidney, and bone raise serum calcium levels.
- A negative feedback loop is in place to control this, driven by FGF-23, hyperphosphatemia and 1,25(OH)2 vitamin D itself.
- This control loop increases 1,25(OH)2 vitamin D breakdown by enzymes, including 24-hydroxylase (24(OH)ase) to water soluble products which can be excreted in the urine, including 1,24,25-trihydroxyvitamin-D.
- 24(OH)ase is part of the P450 enzyme system, and is encoded by the CYP24A1 gene.
Phew! Back to focus on our case. As per Figure 2, in hypercalcaemia with low iPTH you should measure PTHrP, 25-OH vitamin D, and 1,25(OH)2 vitamin D levels. Note that outside of hypercalcaemia or renal dysfunction measuring 1,25(OH)2 vitamin D levels is not recommended).
With hypercalcaemia and elevated 1,25(OH)2 vitamin D levels one should first think of:
- Lymphoma
- Granulomatous disorders (e.g. sarcoidosis, TB)
Why is this?
- Ectopic 1α-hydroxylase produced within a lymphoma or granuloma can lead to excessive 1,25(OH)2 vitamin D generation, resulting in hypercalcaemia
- This 1α-hydroxylase production is not subject to the usual negative feedback control mechanisms.
- In pulmonary granulomas (sarcoidosis and TB), alveolar macrophages are the main source of 1α-hydroxylase expression, which in sarcoidosis can be suppressed by steroid treatment.
- The mechanism of hypercalcaemia in other granulomatous diseases is assumed to be similar.
Interestingly, our patient just had significant sun exposure, and this so-called ‘seasonal hypercalcaemia’ (due to the increased availability of vitamin D precursors) is well-reported in sarcoidosis. However, the lack of pulmonary infiltrates, lymphadenopathy and normal ACE activity makes this unlikely. Hypercalcaemia in TB can be associated with high, normal or low levels of 1,25(OH)2 vitamin D, but again there is no evidence of TB in this case, or of lymphoma.
So where shall we go from here?
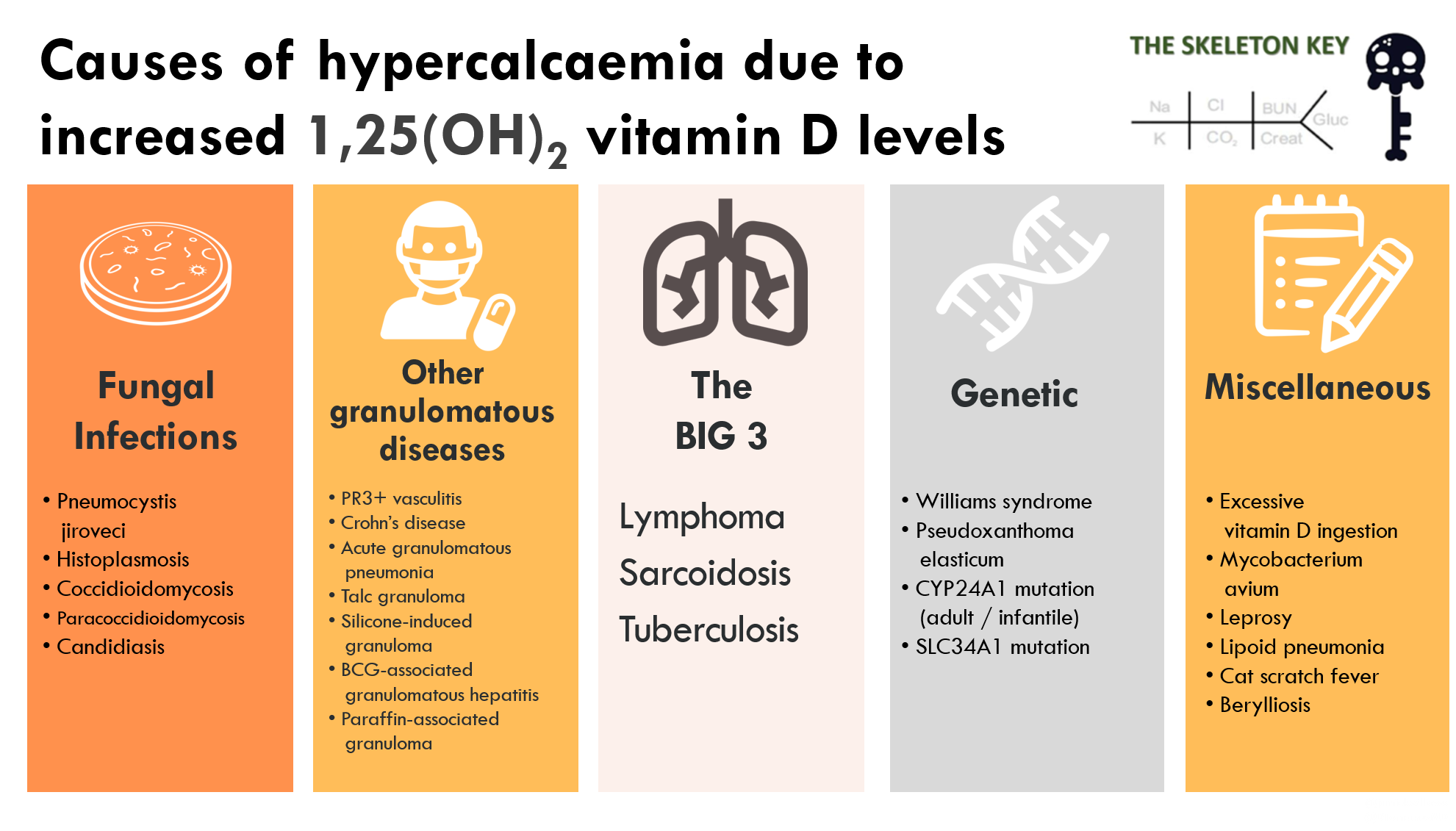
When the admitting team seems to have already done everything you would have thought of (and more!), it can be really intimidating. When that happens, you need your ‘weird causes of hypercalcemia with high activated vitamin D levels’ list saved somewhere handy (Figure 5).
D. More Data
Given the severe presentation without a diagnosis despite extensive investigation, and the history of kidney stones in his sibling, focused genetic studies were performed. These revealed homozygous mutations in CYP24A1, the gene which encodes 24(OH)ase, the key enzyme for breaking down 1,25(OH)2 vitamin D to water soluble products for excretion.
E. The Final Diagnosis and Management
When faced with hypercalcaemia and increased 1,25(OH)2 vitamin D levels, you must first rule out lymphoma and the more common granulomatous diseases like TB and sarcoidosis. After that, the diagnosis is going to be rare.
In this case, the positive family history gave a hint towards a genetic cause and focussed genetic testing was performed. CYP24A1 mutations (Figure 6) leading to hypercalcemia are increasingly recognized with modern genetic sequencing techniques.
- The CYP24A1 mutations cause impaired breakdown of 1,25(OH)2 vitamin D by 24-hydroxylation by 24(OH)ase, in the intestine and kidney.
- This leads to increased levels of 1,25(OH)2 vitamin D, and therefore hypercalcaemia
The hypercalcaemia seen in CYP24A1 mutations is sensitive to vitamin D intake, and the infantile form (idiopathic infantile hypercalcaemia) is often exacerbated by exogenous vitamin D drops. However, adult onset presentations of variable spectrum of severity are increasingly reported, and are also noted to be exacerbated by vitamin D intake or ultraviolet B exposure, just like the ‘seasonal hypercalcaemia’ seen in sarcoidosis.
How is hypercalcaemia in CYP24A1 mutations managed?
Our patient’s kidney dysfunction only partially improved with volume expansion. He was advised:
- conventional calcium stone prevention, which included adequate water intake and low sodium chloride diet
- avoidance of vitamin D products and sunlight exposure
- bone mineral density testing
It was noted that his serum calcium fluctuated over years of follow-up, often corresponding to his level of sunlight exposure.
Addition of the antifungal agent and cytochrome P450 inhibitor fluconazole to inhibit 1α-hydroxylase has also been successfully used, though the long term safety profile of this strategy is unknown.

E. Take Home Points
- Take home points
- Remember the basics in hypercalcaemia – first check intact PTH and if low, check 25-OH vitamin D, 1,25(OH)2 vitamin D and PTHrP levels.
- Hypercalcaemia with raised 1,25(OH)2 vitamin D should raise concern for ectopic 1α-hydroxylase production by lymphoma, sarcoidosis or TB. If ruled out, then review the list of rare causes.
- Genetic diagnoses are increasingly being recognized with sophisticated techniques, and often there will be clues in the family history. Making the diagnosis can open up new treatment strategies, such as the use of fluconazole in CYP24A1 mutations.



Well taught! Thanks
Muito interessante! Muito didático!
SUPERB APPROACH AND TEACHING STYLE
Fantastic! Thanks.