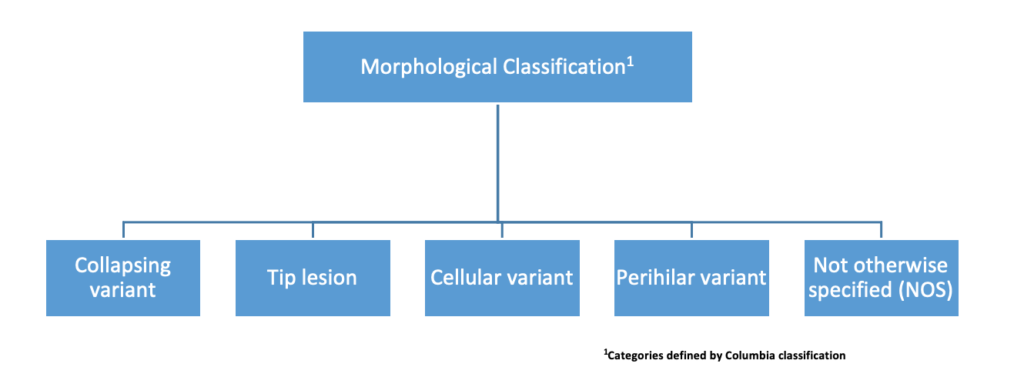
Definition
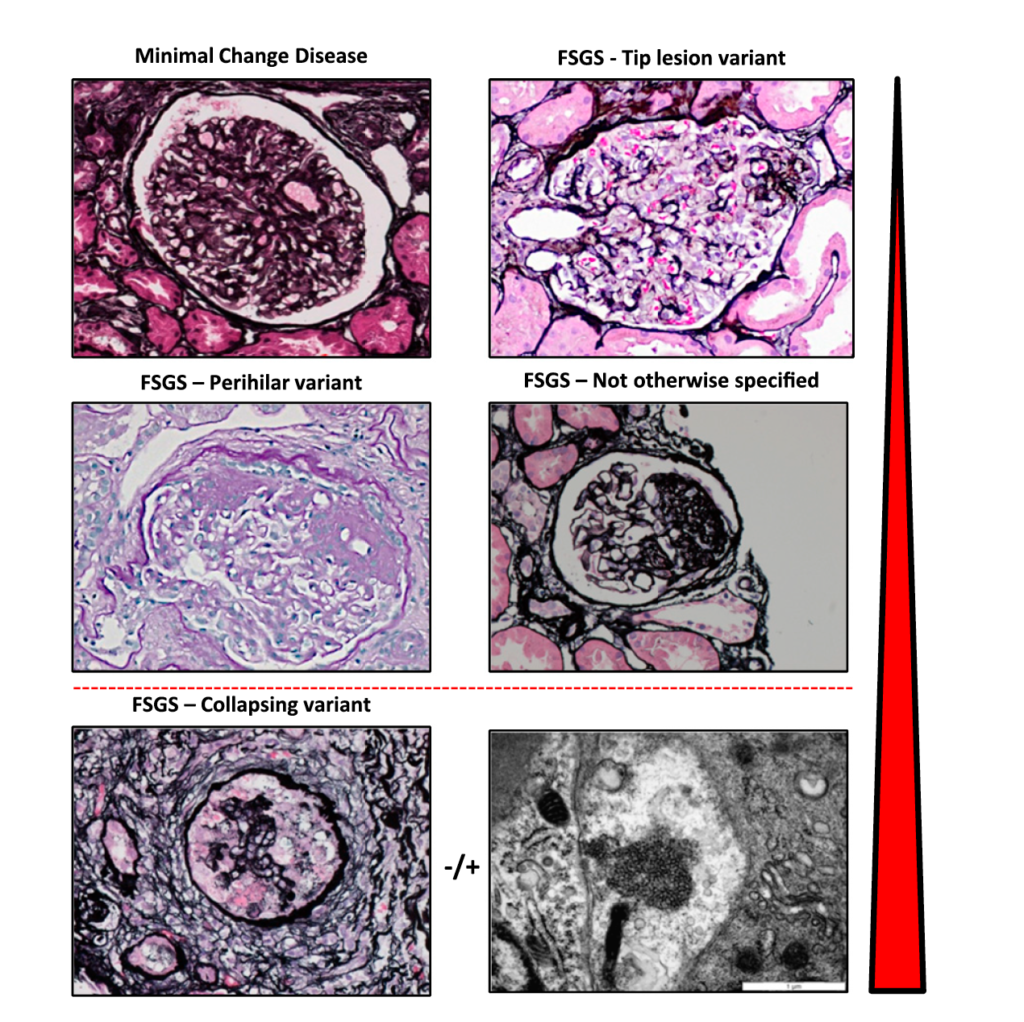
- A group of podocytopathies (of varying etiologies) share a common morphologic feature of focal segmental glomerulosclerosis (FSGS), typically with moderate to heavy proteinuria.
- The term “sclerosis” means healing and it is characterized by accumulation of glomerular collagen (type IV).
- Some of the segmental lesions are not sclerosis, but hyaline deposits or hyalinosis. This feature originated the classical denomination, more of the French school, focal and segmental hyalinosis.
- Other investigators designate those cases in which there are hyaline segments as “FSGS with hyalinosis”.
- There is no a clear explanation why in some cases there are hyaline segments and while none in others. Some authors consider that hyaline lesions are precursory of those sclerosing lesions. Nevertheless, it seems that in many cases the sclerosing lesions begin without a phase of hyalinosis.

Origins and Epidemiology
- Focal and segmental glomerular hyalinization and capillary loop degradation described by Fahr in 1925.
- FSGS was recognized as distinct entity by International Study of Kidney Diseases in Children in 1970s
- Most common cause of nephrotic syndrome in adults
- Apparent increase in incidence over past 2 decades.
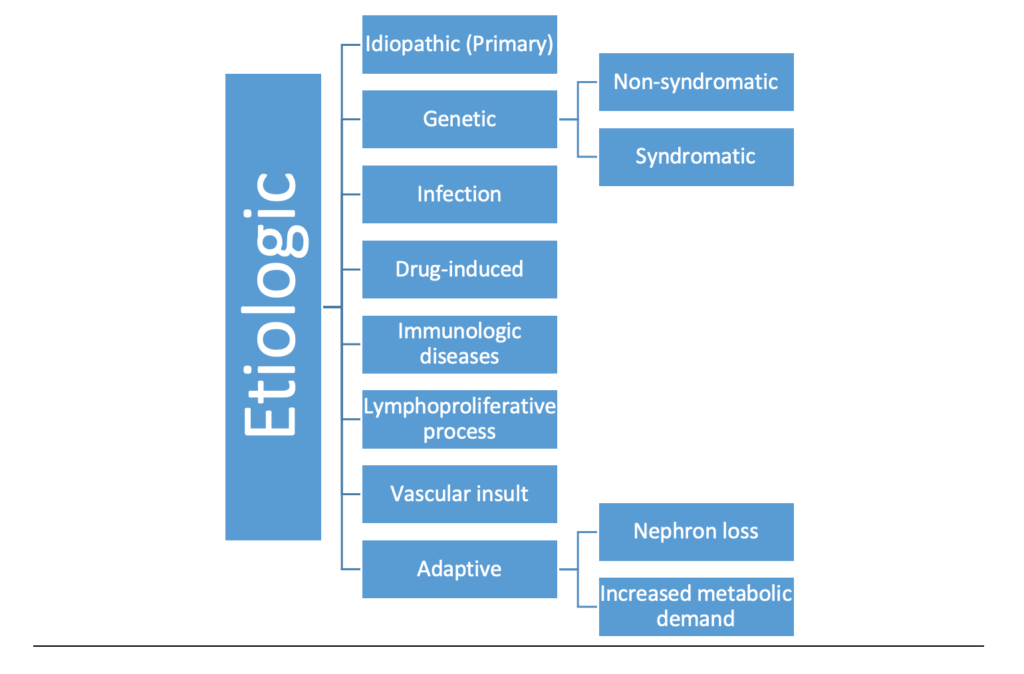
Pathogenesis
- Familial podocytes protein defects (mutations in different proteins like TRPC6, podocin, Nephrin , and WT-1 transcription factor)
- Abnormal cytokines thought to play major role in idiopathic (primary) FSGS development
- Podocyte dysregulation/dysfunction
- Loss of podocytes leads to adhesions
- Risk factor in patients with African descent is APOL1 gene
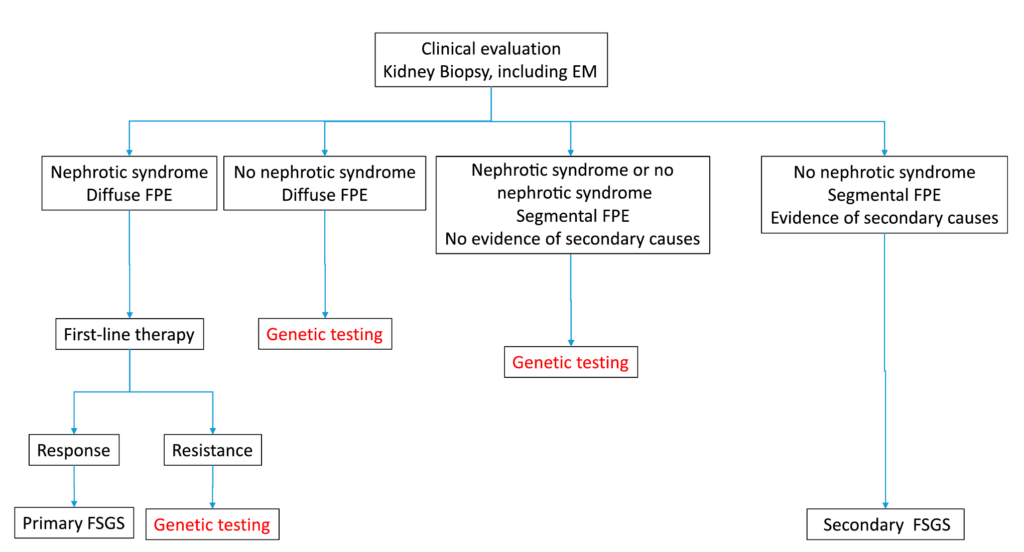
Figure 4. Opinion-based approach to genetic testing in adult-onset FSGS. Note that viral- and drug-associated forms of FSGS are usually excluded by clinical and serologic evaluation. An S. De Vriese et al. JASN 2018.
Pathologic features
Lets begin with features on light microscopy.
Glomeruli
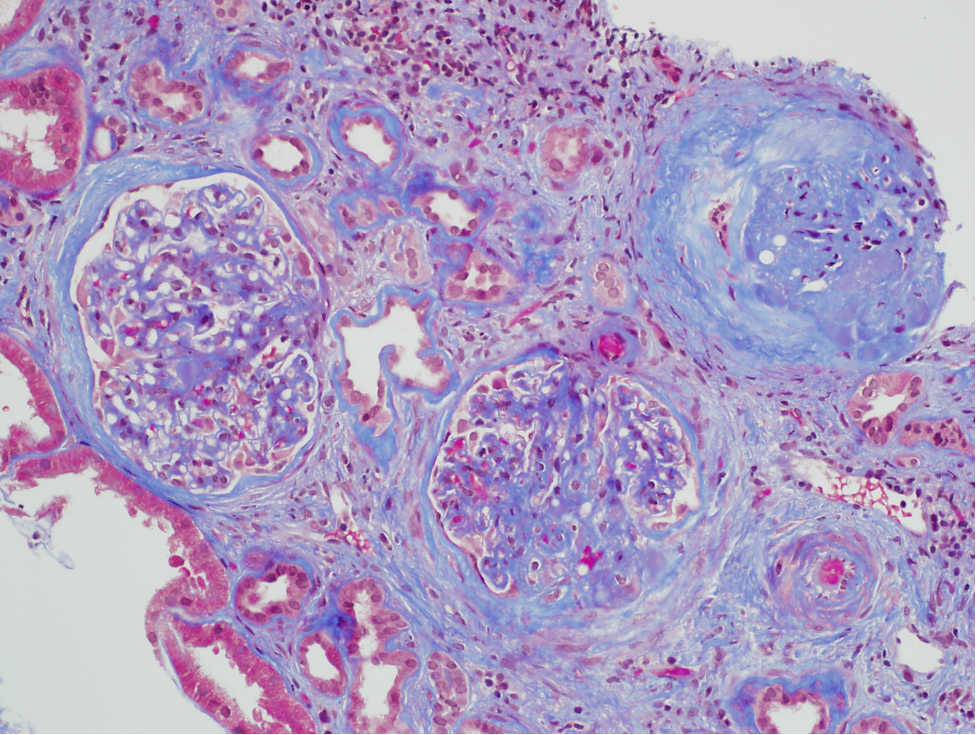
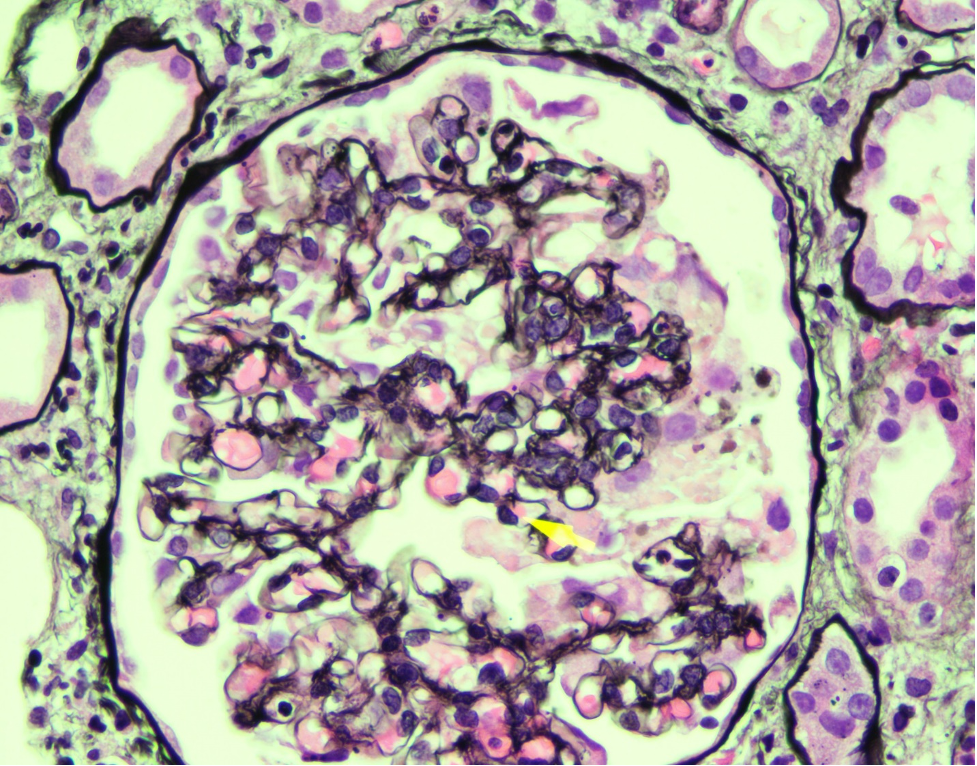
- Sclerosis involves some glomeruli (focal) and only portion of glomerular tuft (segmental). Of note, global sclerosis may be incidental finding and is not particularly useful in making diagnosis of FSGS (image 1).
- Adhesions (synechiae) of glomerular tuft to Bowman space often accompany segmental sclerosis and are often seen early in sclerosis process.
- Hyalinosis.
- FSGS has zonal distribution, beginning in corticomedullary (juxtamedullary) junction (CMJ)
- Glomerular hypertrophy often accompanies FSGS
- Adequate sample of glomeruli is crucial to make diagnosis of FSGS since focal lesions may be missed with small samples
- Multiple levels (step sections) may be needed to identify focal segmental lesions
Tubules
- Tubular epithelial cells contain PAS(+) reabsorption droplets due to glomerular proteinuria.
- Tubular atrophy is typically only focal early in course of FSGS.
- Tubulointerstitial changes pronounced in collapsing variants of FSGS and in HIVAN.
Interstitium
- Interstitial fibrosis (IF) may be present and is typically focal.
- Interstitial inflammation is absent or minimal.
Vessels
- Arteriolar hyalinosis and arterial intimal fibrosis may be prominent late in course of FSGS.
Immunofluorescence
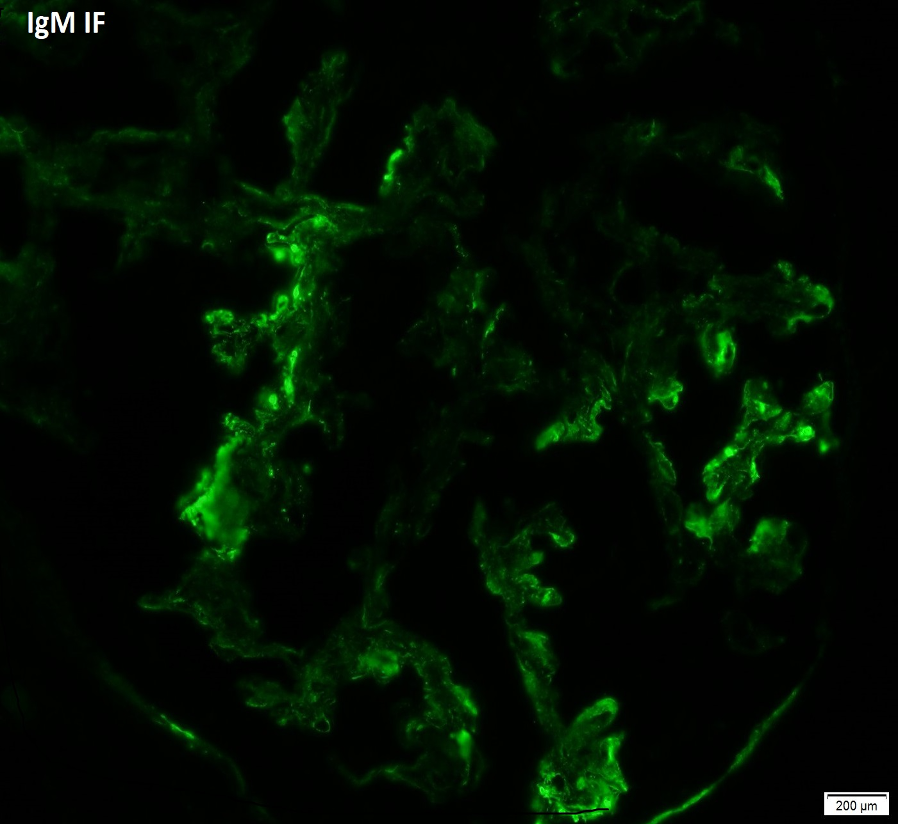
- IgM and C3 often positive in sclerotic areas or areas of increased mesangial matrix (image 4).
Electron Microscopy (Image 4)
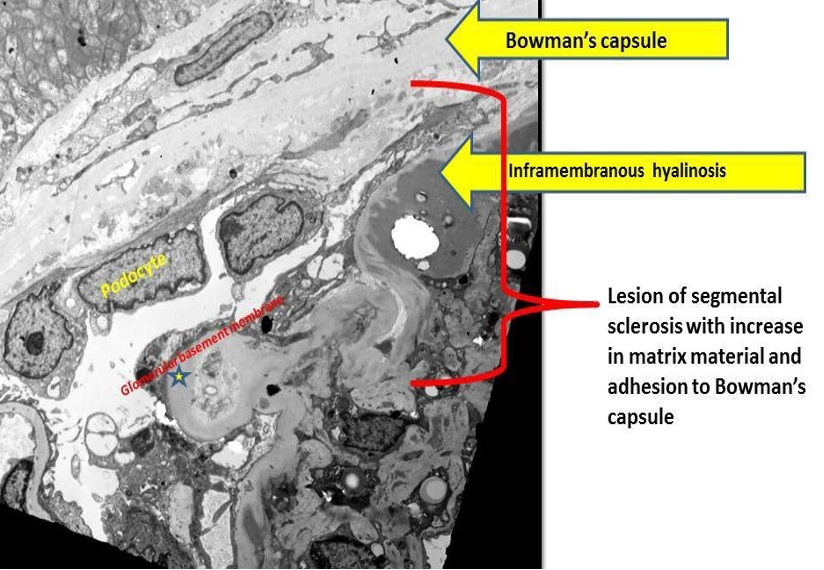
- Foot process effacement (FPE) typically not complete, less
- Secondary FSGS usually shows less FPE than idiopathic forms of FSGS
- In HIVAN, tubuloreticular inclusions can sometimes be identified
- Subepithelial multilamination of GBM in collapsing variant
Differential diagnosis
- Focal Glomerulonephritis
- IgA nephropathy, lupus, and other inflammatory glomerulonephritis can result in segmental scars in glomeruli
- Breaks in GBM sometimes evident in PAS/silver stains (not seen in FSGS)
- Minimal Change Disease
- No segmental sclerosis, unless segmental sclerosis is preexistent due to other etiology
- FPE is usually diffuse in MCD but is typically less in most cases of FSGS
- Numerous globally sclerotic glomeruli and vascular changes in patient with nephrotic syndrome are suggestive of FSGS even if no segmental lesions are seen
- Difference in outcome between MCD and FSGS equivalent to difference between benign and malignant
Sam Albadri
Hennepin Hospital
Minnesota, USA


