In the recent edition of Hepatology, the International Club of Ascites (ICA) have published their revised recommendations on the diagnosis and management of AKI in patients with cirrhosis. They first proposed diagnostic criteria for Hepatorenal syndrome (HRS) in 1996 and last updated them in 2007 (covered by RFN). Since their first publication, there has been much work done in defining AKI with RIFLE, AKIN and KDIGO and this is reflected in the ICA’s latest guidance. This time, they have gone beyond defining HRS to include staging of AKI in patients with cirrhosis and proposed how this could be used to inform management.
Here are the definitions they propose (the ICA-AKI classification):
The first thing to note is that they’ve removed the fixed threshold of sCr of ≥ 1.5 mg/dl (133 mmol/l) from the old criteria and brought in an absolute increase in sCr. Indeed, the eagle eyed amongst you will have noticed that their definitions and staging of AKI are based on the KDIGO criteria but with the use of urine output removed. The rationale for this being that patients with cirrhosis can often be oliguric with avid sodium retention but still have preserved renal function. Conversely, they can have an increased urine output as a result of diuretic therapy.
Whilst a sCr less than 7 days before admission would be the ideal result to use as baseline, they have pragmatically suggested that the most recent value available in the last 3 months can be used. When this is not available, the admission sCr should be used as baseline. They do not recommend using the reverse application of the MDRD formula to calculate an estimate of baseline sCr as MDRD is known to be inaccurate in patients in cirrhosis (covered in a previous RFN post ). If no previous sCr is available and the admission value is ≥ 1.5 mg/dl (133 mmol/l), decisions should be based on clinical judgement.
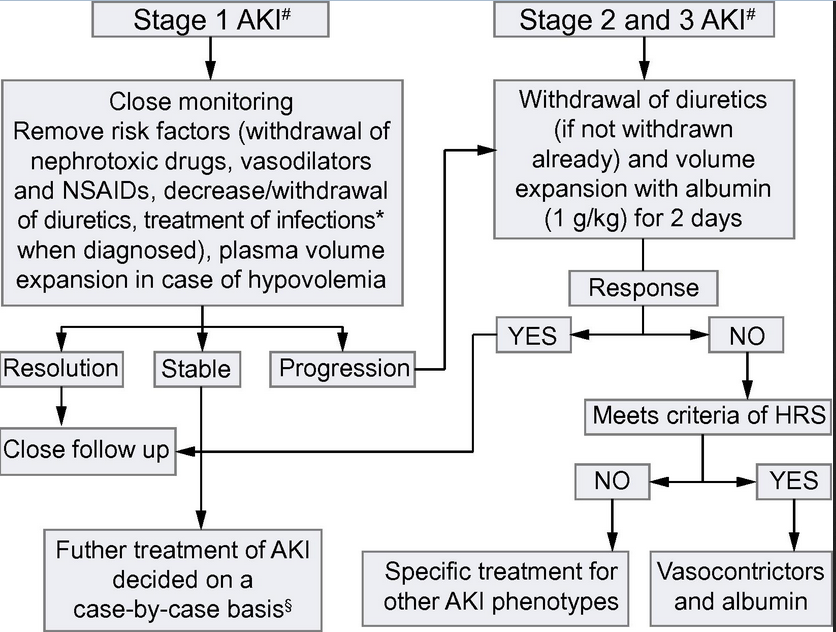
Moving onto management, the ICA has suggested the following algorithm:
Treatment for those with AKI stage 1 is outlined in the table. Note that the plasma volume expansion can be with crystalloid or albumin (or blood in the case of GI bleeding) at this stage. Those who present with AKI stage 1 who progress are then treated in the same way as patients who present with AKI stage 2 and 3 i.e. stopping the diuretics and 48 hours of albumin 1g/kg (max dose 100g per day). Further management then varies on the aetiology and whether or not this is HRS, which the algorithm helpfully informs. The new criteria for HRS:
Again, they have removed the cut off value in sCr from the diagnosis in favour of a dynamic change in sCr as per the ICA-AKI criteria. Importantly, they have suggested revising the diagnostic criteria for type 1 HRS and with it the indications for treatment with vasoconstrictors. Type 1 HRS is currently defined as a doubling of the initial sCr to level ≥ 2.5mg/dl (226 mmol/l) in < 2 weeks. Evidence however suggests that a higher sCr at the beginning of treatment leads to a lower chance of response to vasoconstrictors and albumin. With the new algorithm, patients at AKI stage 2 or 3 or who have progression from stage 1 that meet all the other criteria for HRS, should receive treatment with vasopressors irrespective of the final value of sCr. This will allow treatment earlier and hopefully lead to better outcomes though at present there is no data to confirm this.
No consensus was reached on the optimal management of those patients who present with AKI stage 1 but then have stable renal function (i.e. do not regress or progress). Whilst all agreed that if the patient has a final sCr ≥ 1.5 mg/dl (133 mmol/l), they should be treated according to the right side of the algorithm, most of them had concerns about the use of vasoconstrictors for those with HRS if the sCr was < 1.5 mg/dl (133 mmol/l). Hence, they advise to treat these patients on a case-by-case basis.
Authored by David Baird,
Royal Infirmary of Edinburgh