Previously, we discussed the sonographic anatomy and principles of image acquisition of the apical view. Now let us talk briefly about some of the important abnormalities that can be seen on this view.
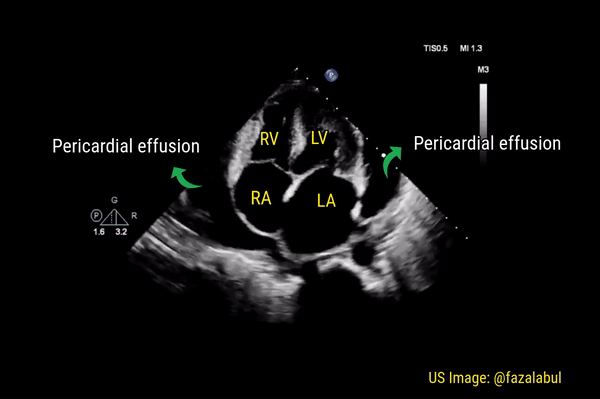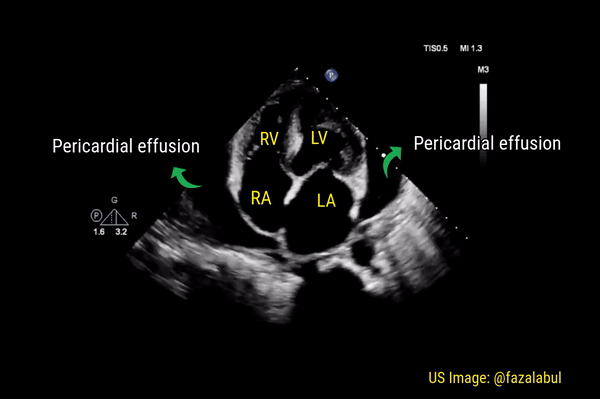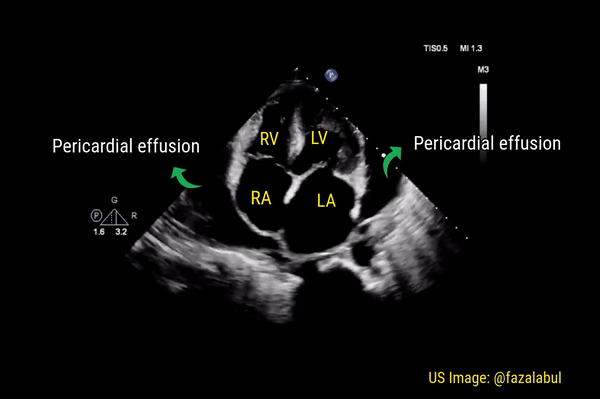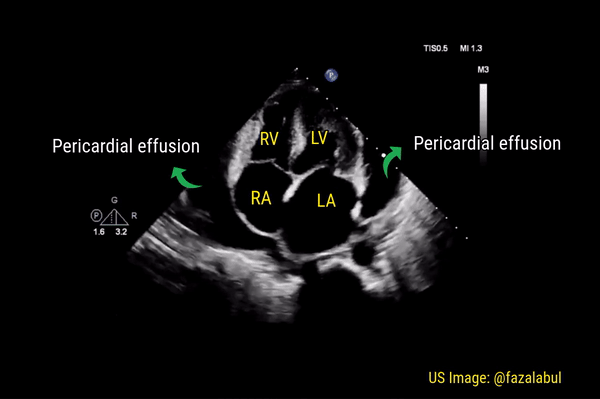
Pericardial effusion
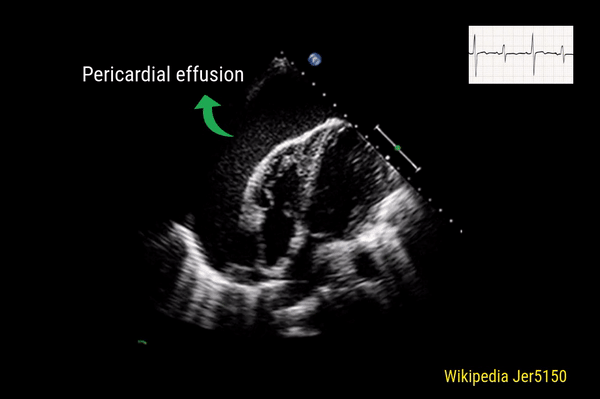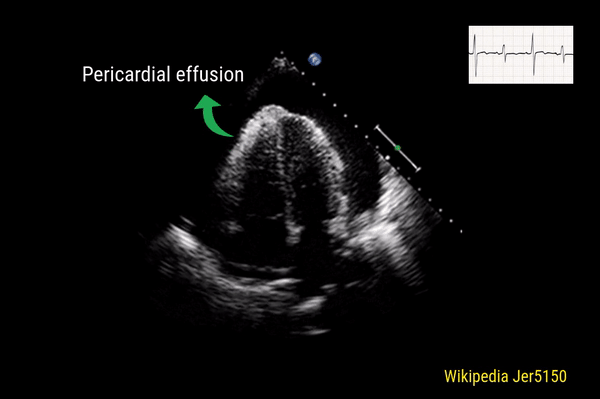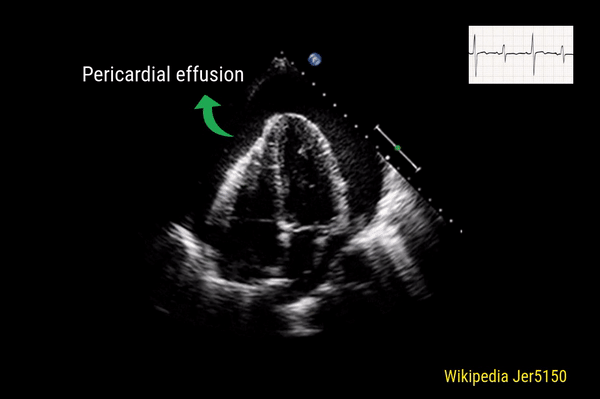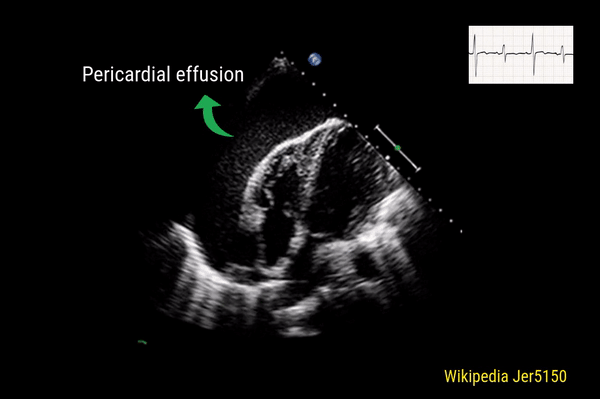
This is a good view to visualize the extent of pericardial effusion (circumferential vs localized) as well as comment on the right sided chamber collapse. As mentioned before, pericardial effusion appears as an anechoic or echo-free space between the two layers of the pericardium. Figure 1 demonstrates a large circumferential pericardial effusion. In general, size of the effusion is assessed by the greatest width of the anechoic space in ‘diastole’: small = less than 1 cm wide, moderate = 1-2 cm and large = more than 2 cm. Figure 2 is another large effusion with swinging heart, the echocardiographic equivalent of pulsus alternans. Note diastolic right atrial collapse (diastolic = when AV valves open) suggestive of tamponade physiology.
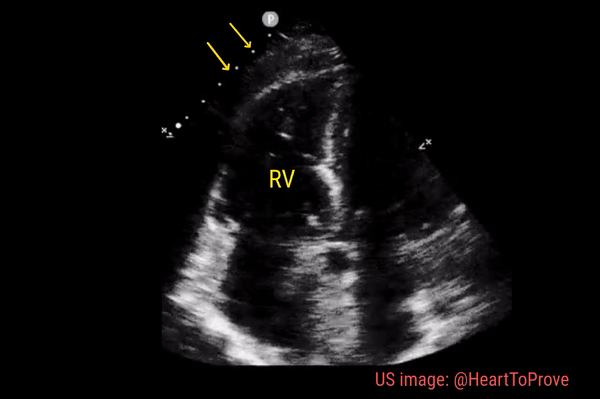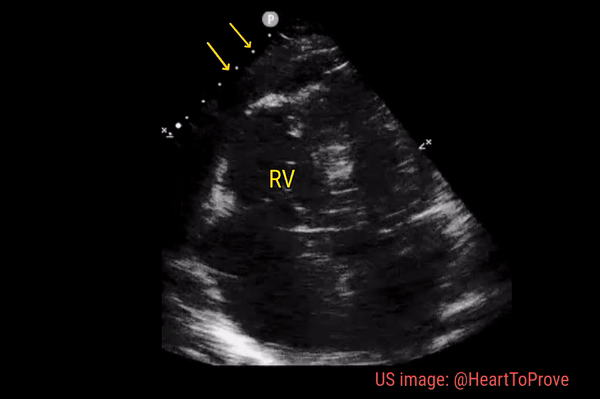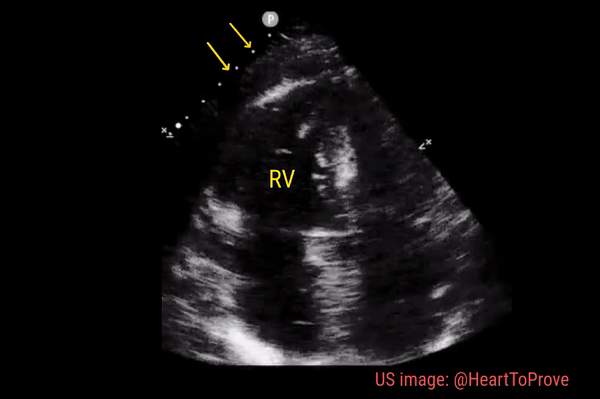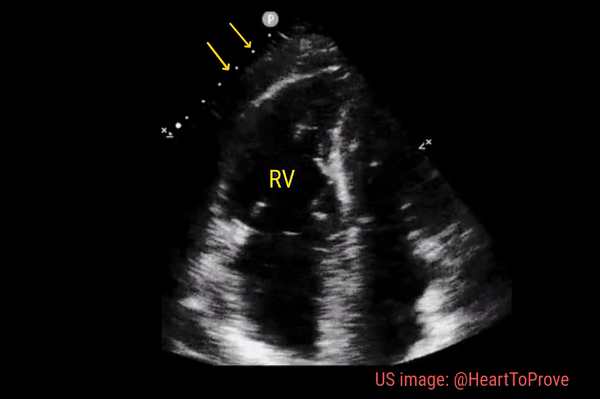
Dilated right ventricle
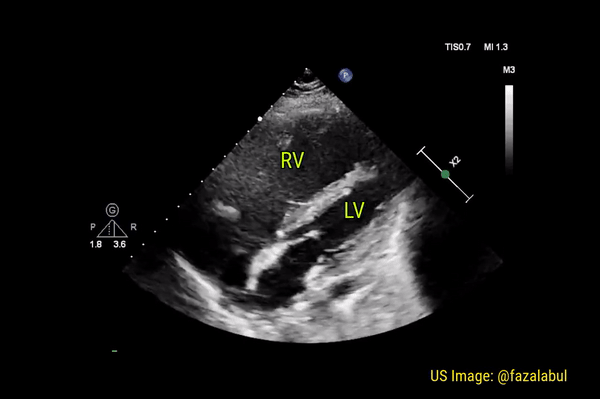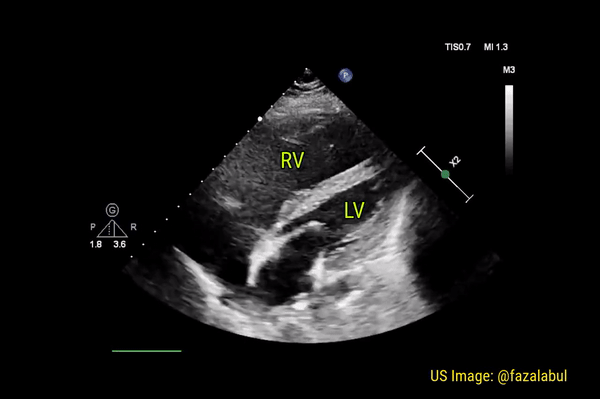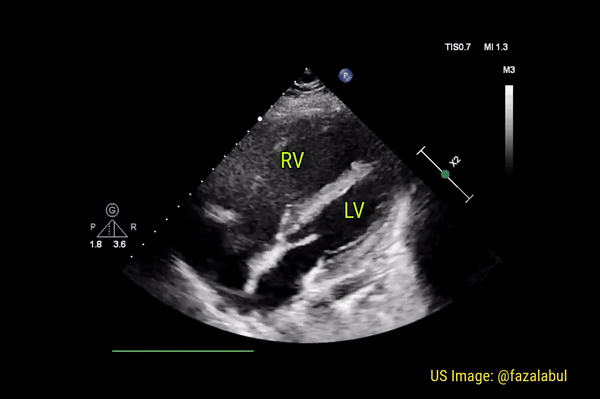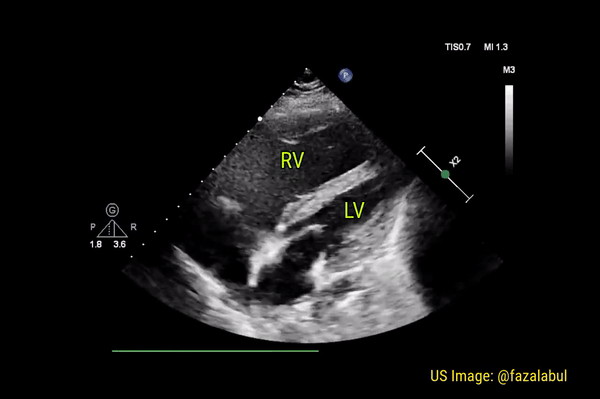
Normally, the right ventricle is triangular in shape with a cavity up to two-thirds of the left ventricle. Figure 3 demonstrates a case of severely dilated right ventricle (RV size more than that of LV) in a patient with severe pulmonary hypertension. If you obtain PSAX view in this patient, you will most likely see a D-shaped left ventricle due to flattening of the interventricular septum. We have to be very cautious in administering fluids to these patients because further increase in right ventricular size (due to fluid) coupled with obstruction to outflow (due to pulmonary hypertension) results in additional pressure on the septum leading to decrease in cardiac output and cardiogenic shock.
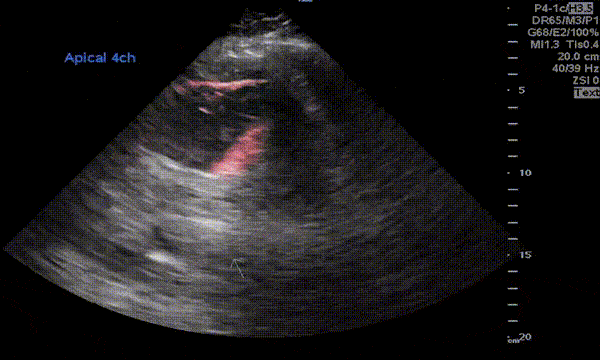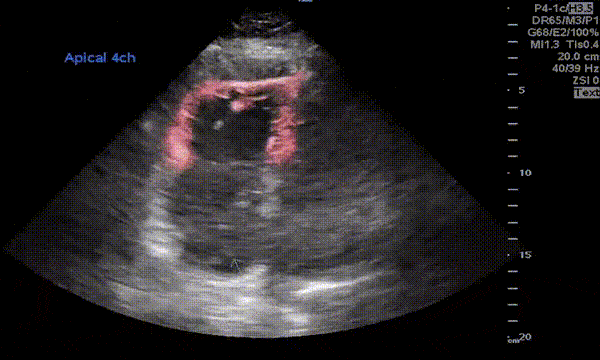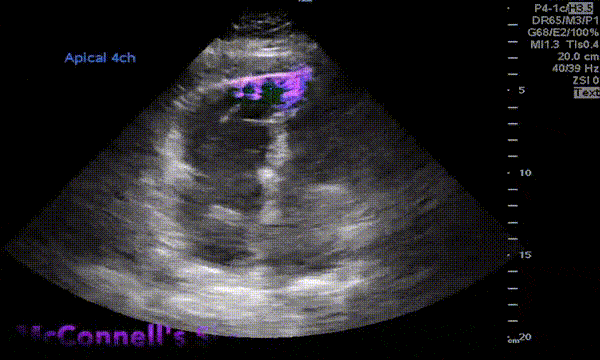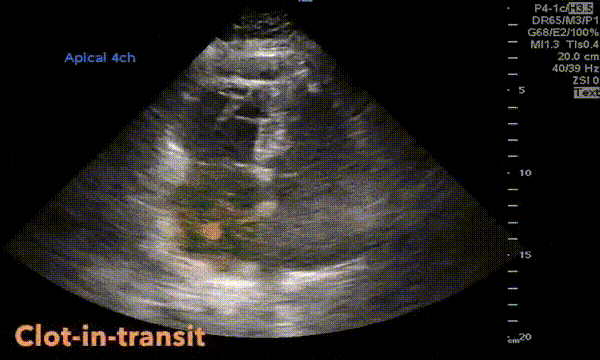
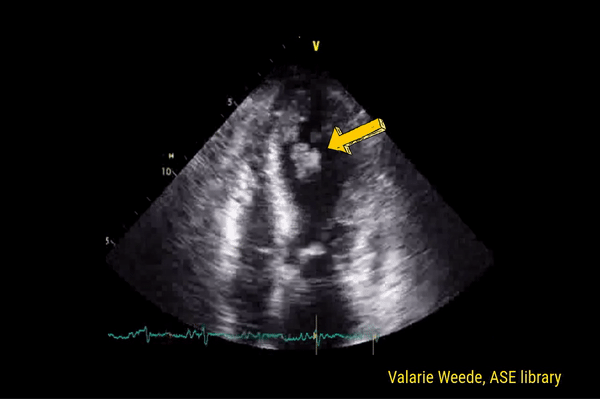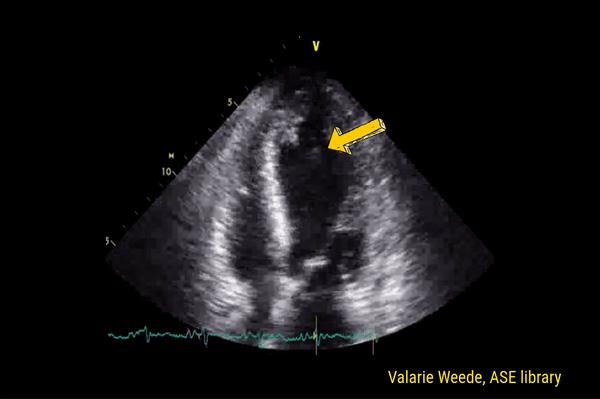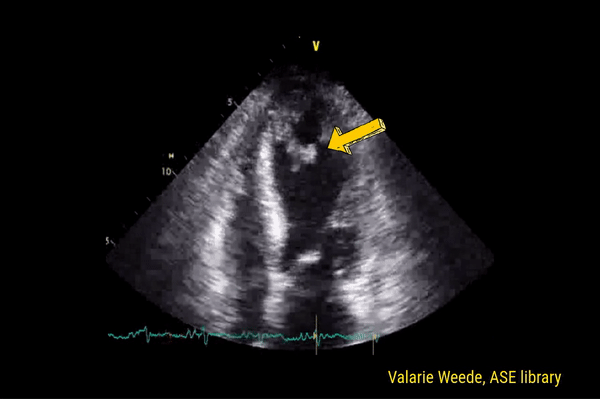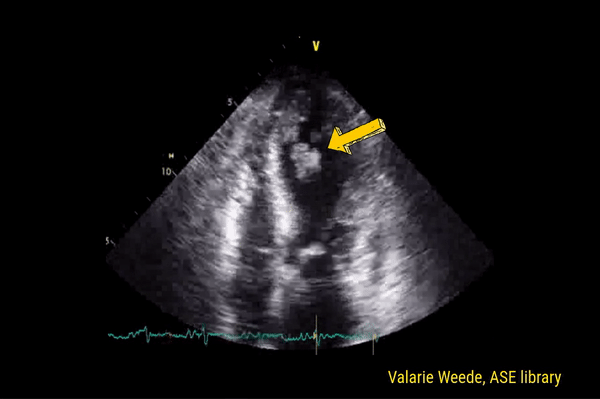
McConnell’s sign
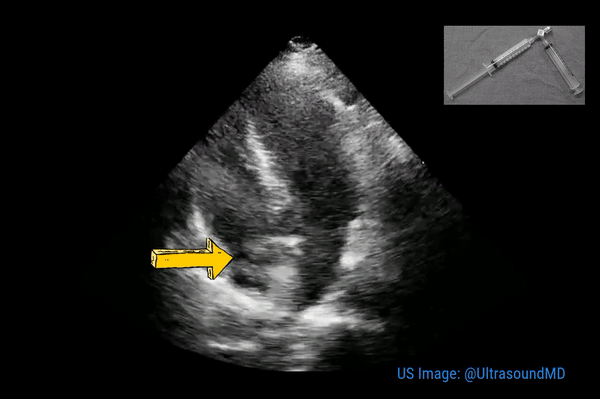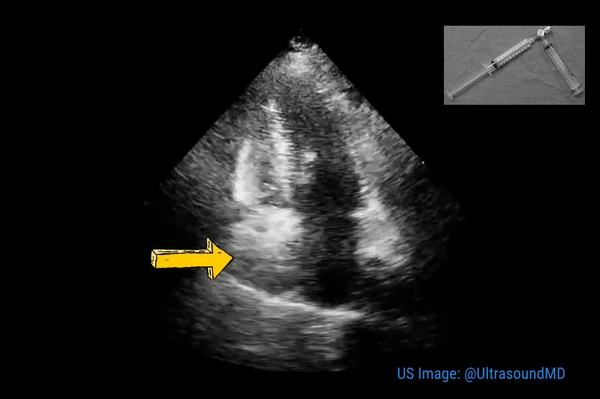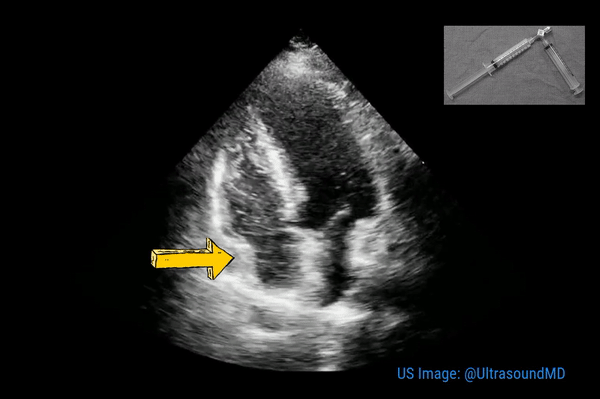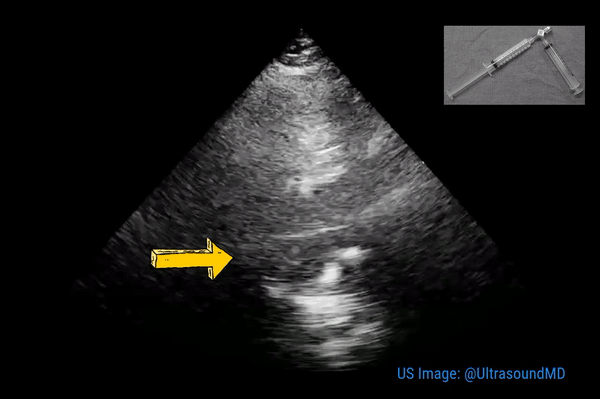
Described in 1996 by McConnell et al, this sign is indicative of acute right ventricular strain typically seen in pulmonary embolism. It is characterized by hypokinesis of the RV free wall with preserved contractility of apex (Figure 4). Though can be seen in RV myocardial infarction and chronic pulmonary hypertension, McConnell’s sign is considered to be fairly specific for acute pulmonary embolism in the appropriate clinical context. For example, when a patient with nephrotic syndrome or antiphospholipid antibody syndrome complains of shortness of breath, look for right ventricular strain, especially when the lung ultrasound does not suggest significant congestion. You may also see a clot in transit appearing as a mobile echogenic material in the right atrium or ventricle in this setting, associated with high risk of mortality. Figure 5 is an excellent illustration of McConnell’s sign, dilated right ventricle and clot in transit in a patient with acute pulmonary embolism (courtesy: @eabramsMD).
The rapid atrial swirl sign
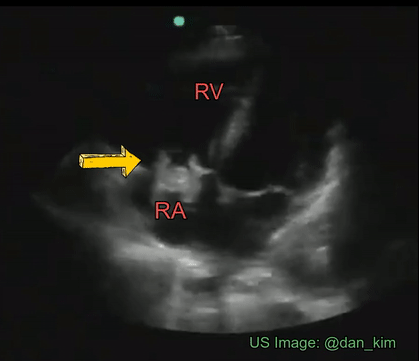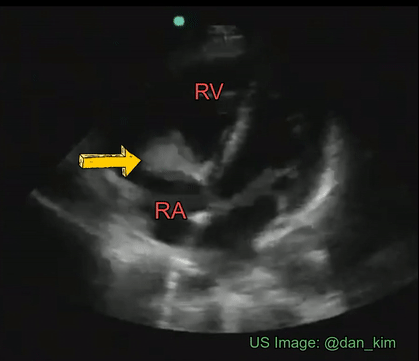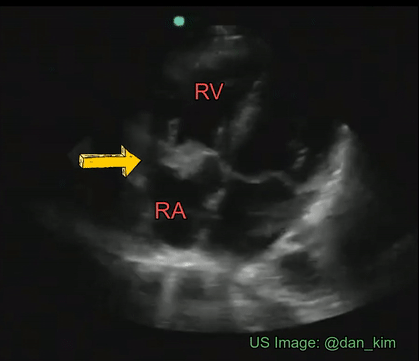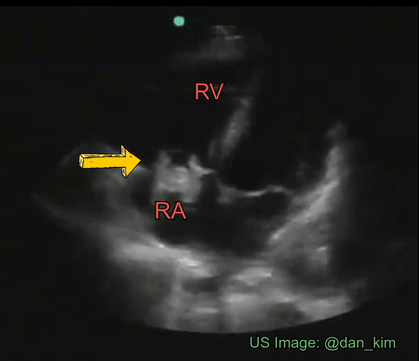
After placing an internal jugular hemodialysis catheter, we normally wait for a chest X-ray to confirm the tip position. Instead, we can look for a positive “atrial swirl” on POCUS as soon as the line is placed and use it immediately. It involves injecting either a regular 10 cc saline flush or agitated saline and watching for opacification of the right atrium in the apical 4 chamber or subcostal views. Normally, you should see the opacification almost immediately (less than 2 seconds), which means the line placement is appropriate and is usable. If the swirl is delayed or absent, obtain a chest X-ray to evaluate for misplacement of the line. Figure 6 demonstrates this sign.
Takotsubo cardiomyopathy:
Occasionally, you may encounter Takotsubo cardiomyopathy while evaluating a patient with shortness of breath. It is characterized by systolic apical ballooning of the left ventricle due to depressed movement of mid and apical segments and hyperkinesis of the basal segments. It is kind of reverse McConnell’s sign on the left. The name is derived from the Japanese word “tako-tsubo” meaning “fishing pot for trapping octopus” based on the appearance of the ballooned LV (Figure 7). As it is typically precipitated by acute emotional stress, Takotsubo cardiomyopathy is also called “stress cardiomyopathy” or the “broken heart syndrome”. Acute myocardial infarction (LAD lesion) can be associated with similar echocardiographic findings and therefore immediate cardiology consultation should be obtained for possible coronary angiography whenever you see this pattern on FoCUS.
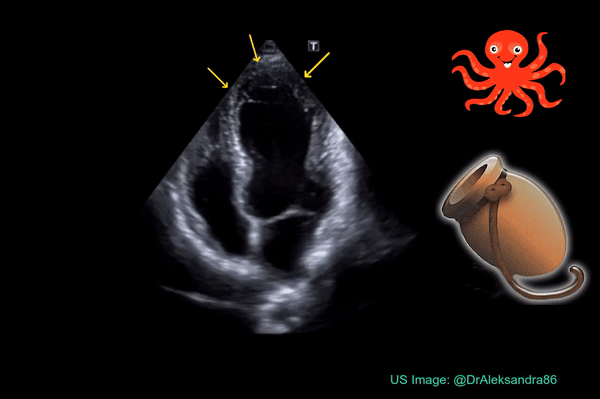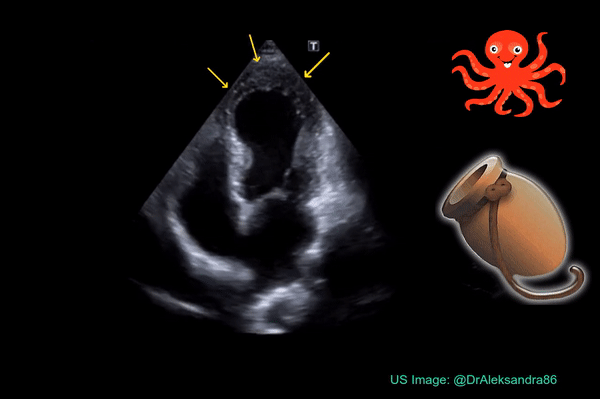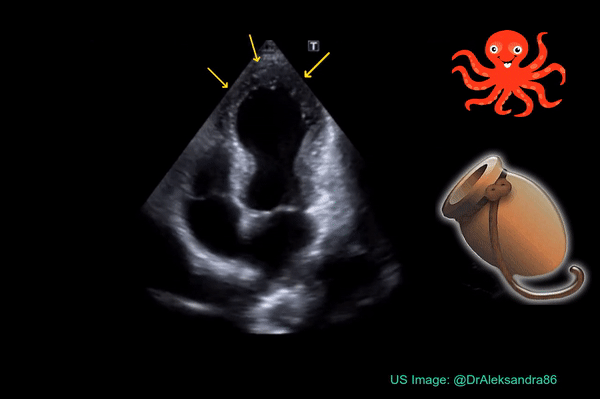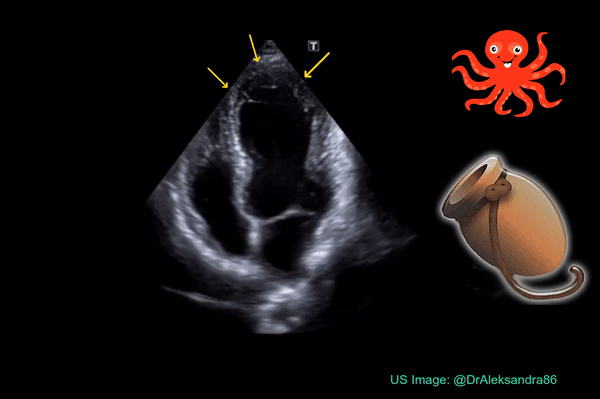
Left ventricular thrombus
This is one of the important incidental findings on FoCUS that nephrologists need to be aware of as we often take care of patients with heart disease. LV thrombus, which appears as a mobile echogenic mass is a well‐known complication in the course of acute myocardial infarction and can be associated with thromboembolic events. Figure 8 represents one such case. Note that there is apical ballooning similar to that of Takotsubo but this patient had acute LAD territory infarction. Interestingly, LV thrombi are relatively rare among patients with Takotsubo cardiomyopathy.
Valvular vegetation
Though transesophageal echocardiography is required in most patients suspected to have infective endocarditis because of better sensitivity, valvular vegetations can sometimes be seen on a transthoracic echocardiogram. It is important for the nephrologists to be aware of the characteristic features of these lesions as we frequently take care of patients with dialysis access infection and bacteremia. Essentially, vegetation is an infected mass usually attached to cardiac valves or implanted intracardiac materials such as pacemaker wires. Vegetations tend to be irregular in shape, mobile but attached to the upstream (= low-pressure) side of the valve. Valvular regurgitation due to destructive valve lesions is a frequent accompaniment and hence Color Doppler evaluation should always be performed. Figure 9 demonstrates a tricuspid valve vegetation in a patient who was admitted for fever and suspected pneumonia.
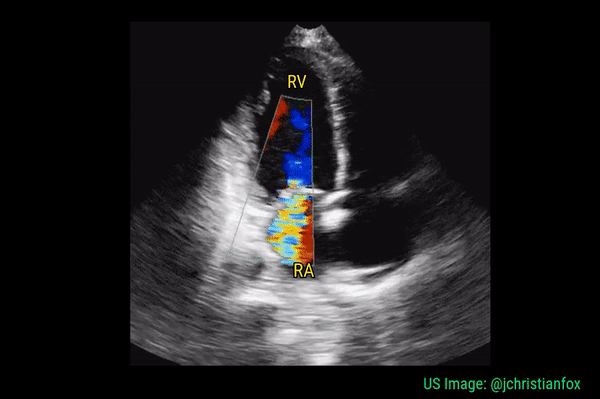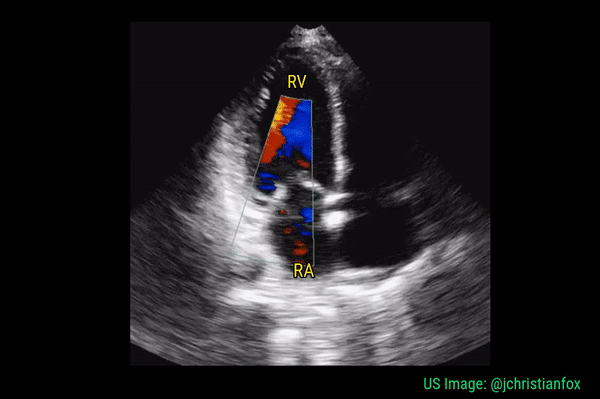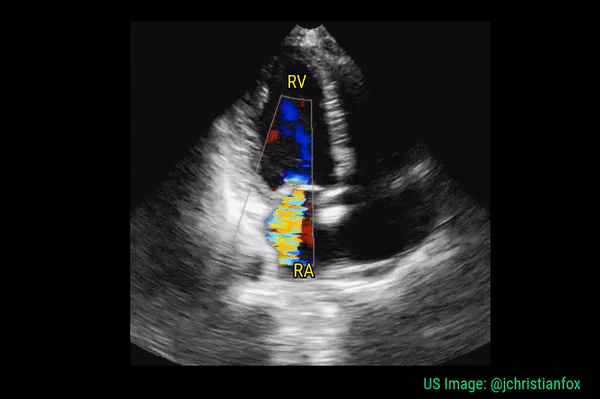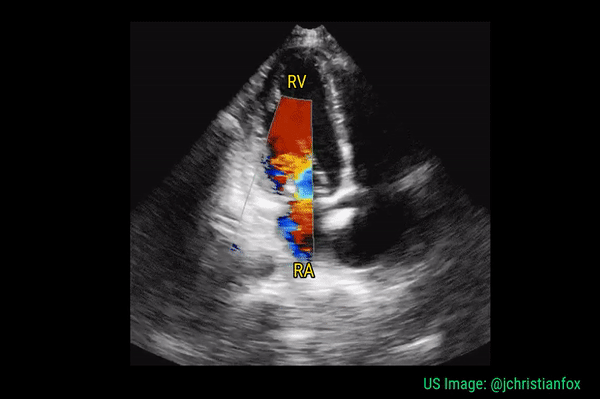
Valvular regurgitation
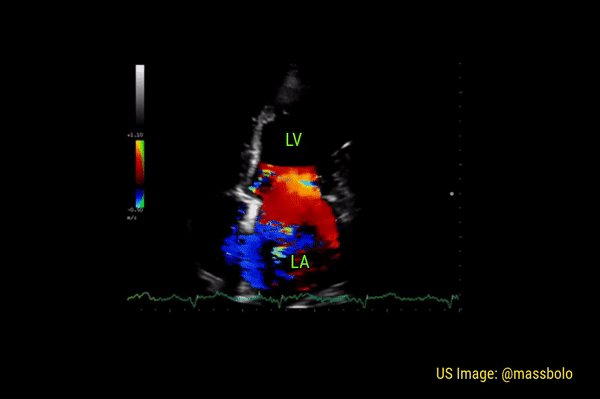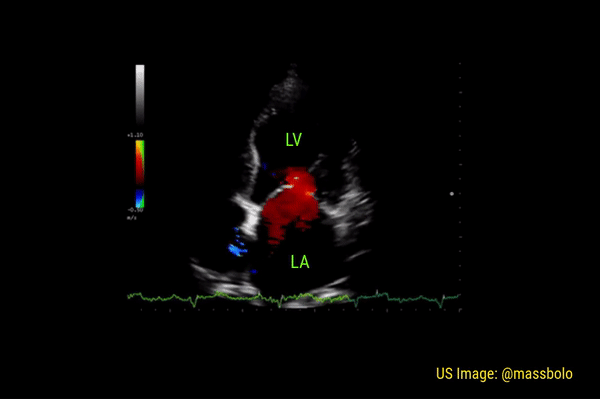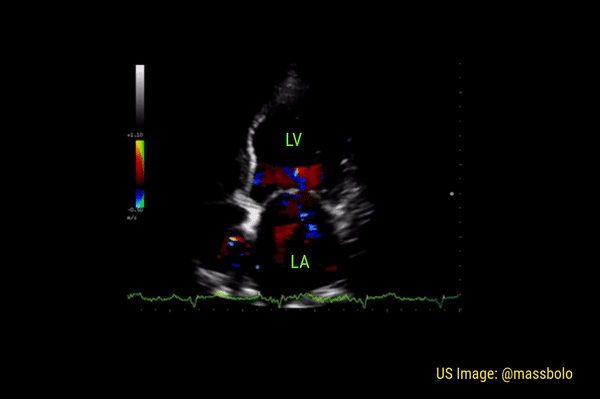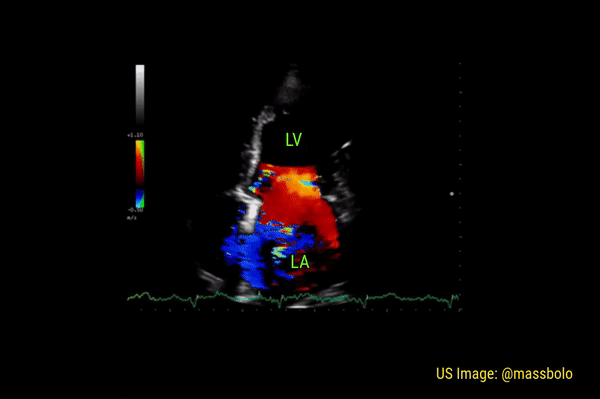
Significant regurgitant lesions of mitral and tricuspid valves can be qualitatively assessed on the apical 4 chamber view using color Doppler. Make sure to use the smallest color box that reasonably includes the valve and the chamber receiving the regurgitant jet. In general, regurgitation of the AV valves is considered severe if the jet (turbulent flow with mixed colors) occupies more than 50% of the atrium (LA in case of mitral valve and RA in case of tricuspid). Figures 10 and 11 demonstrate severe mitral and tricuspid regurgitation respectively. Quantitative evaluation of these lesions using spectral Doppler is beyond the scope of FoCUS.



These are beautiful examples, thank you Dr. Koratala!