The concept of removing excess fluid via ultrafiltration (UF) is not a new one. The first described use of UF dates back to 1974. Back then, drip clamps were used to calibrate the amount of UF. For this experiment, 5 patients underwent UF while also undergoing intermittent hemodialysis (iHD), but the authors suggested that UF could be used in patients that did not require hemodialysis as well. The role of UF in patients with other refractory edematous states such as heart failure has been well-studied since then. The hypothesis was that UF may be able to remove excess sodium and water more efficiently and rapidly as compared to intravenous (IV) diuretics.
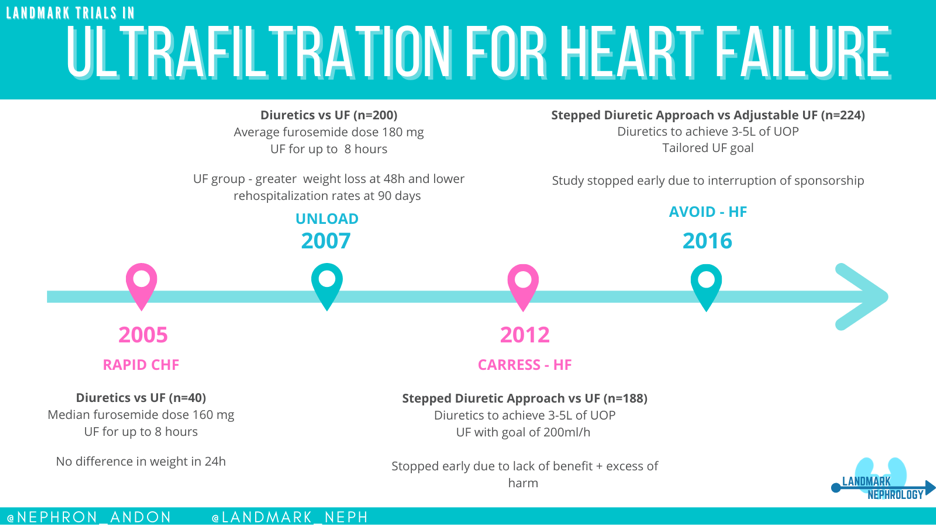
RAPID-CHF aimed to establish the safety of UF use in patients with chronic heart failure and whether it could achieve more rapid fluid removal than that with diuretic use alone. 40 patients were divided in 2 groups and assigned to either diuretics only or a combination of UF with diuretics. The primary end point was weight loss achieved at 24 hours. The UF group received a single 8 hours session of UF. The average creatinine values were 1.6 mg/dL and 1.8 mg/dL in the UF and the diuretic-only group, respectively. At 24 hours, weight loss was 2.5 kg and 1.8 kg in the UF and diuretic-only groups, respectively, but this difference was not statistically significant. The median diuretic dose for the diuretic only group was 160 mg which some may consider to be conservative dosing. One patient developed a catheter-associated infection in the UF cohort but otherwise UF was generally well-tolerated without serious complications. The authors argued that the small sample size limits the ability to draw definitive conclusions about the potential clinical impact of UF versus usual care.
Next came UNLOAD, a larger study that enrolled 200 patients. This trial again compared UF to intravenous diuretics alone but this time, the primary endpoints were weight loss and dyspnea after 48 hours. Safety end points included changes in renal function, electrolytes, and blood pressure. The mean creatinine was 1.5mg/dL at enrollment in both groups. UF treatments were conducted up to 8 hours at a time. After the first 48 hours, greater weight loss was achieved in the UF group with 5.0 vs 3.1 kg loss in the diuretics-only group (p=0.001). The 90-day re-hospitalization rate was also lower in the UF group. With regard to adverse events, one case of catheter-related infection was reported along with one patient developing renal failure in the UF group. In the diuretic-only group, the average furosemide-equivalent daily dose was 180mg, again arguably conservative.
However, the results of the CARRESS-HF trial were not as encouraging. In this trial, patients were randomized either UF only or to a “stepped pharmacology therapy” (i.e. flexible dosage as necessary to achieve as necessary to maintain a urine output of 3 to 5 liters per day). The UF rate goal was 200 ml/h of fluid removal. The original plan was to randomize 200 patients; however, after enrollment of 188 patients, the study was stopped due to lack of benefit and an excess of adverse events observed with UF (72% vs 57% in the pharmacologic-therapy group; p=0.03). The primary endpoints were change in weight and serum creatinine at 96 hours from enrollment. The UF group had a statistically significant worsening in creatinine when compared to the pharmacologic-therapy group and the change in weight was similar between the two groups. The rate of death or rehospitalization did not significantly differ between groups.
Critics of CARRESS-HF propose that the predetermined rate of filtration during UF and possible delayed enrollment of some patients (the average time from admission to enrollment was 34 hours) may have skewed the results. It is also important to note that in this trial patients had a higher initial creatinine (2.0mg/dL) than in previous studies.
To try to overcome these critiques, the AVOID-HF trial was designed to compare adjustable UF rates versus diuretic treatment. This was intended to be one of the larger randomized trials to address this question with a planned enrollment of 810 patients to be randomized within the first 24 hours of admission. The primary outcome here was time to heart failure event within 90 days of discharge. The study was ultimately terminated early by the sponsor due to slower than expected enrollment with a final number of only 224 patients. The results from the trial showed a trend towards longer time free from heart failure event in the UF group but this was not significant. In addition, serious adverse events deemed to be related to study therapy occurred in a higher number of patients in the UF group when compared to the diuretic group.
In summary, the currently available data would suggest that there is potentially greater risk than benefit when using UF as a strategy for volume management in patients without refractory volume overload, advanced renal failure or another indication for dialysis. For these lower risk patients, diuretics appear to be safer. Unanswered questions are how best to manage higher risk patients and whether early dialysis initiation has a role beyond intensive cardiology care with best medical or device therapies. Currently, the American College of Cardiology does not recommend UF as a first line treatment for heart failure.
Post By: Larissa Kruger, MD @nephron_andon
Reviewed by: John Danziger, MD



The CARRESS-HF was an unfortunate rather ill conceived trial in which diuretic treatment was compared with ultrafiltration. This is more or less like comparing gentamycin with ampicillin in the treatment of infection, and as such it did not make too much sense. Diuretics are, for many reasons, the first approach to treat the fluid overload associated with CHF. The use of ultrafiltration, on the other hand, should be reserved to those cases in which diuretics are failing, and simply increasing the diuretic dose worsens azotemia, with little or no relief of fluid overload. Typically, those are cases in which the inferior vena cava is dilated and does collapse. In those case, the use of diuretics cannot increase the pressure of the renal veins to overcome the already elevated pressure in the IVC. There is a proper use of diuretics and a proper use of ultrafiltration, and comparing one with the other is simply wrong. Physicians need to distinguish between conditions in which either of those is the right therapy to use.
These issues require a new better trial, which we are unlikely to see any time soon.
The main issue in heart failure with reduced EF is decreased effective arterial blood volume due to low cardiac output. Excessive fluid removal, whether by diuresis or ultrafiltration, would be more harmful causing more target organ hypoperfusion.
Even in patient with decompensated pulmonary edema we will not usually need to remove 3 liters to improve oxygenation and stabilize the patient’s hemodynamics.
A cautious fluid removal with improving cardiac output and monitoring target organ is the most effective way to manage these cases.
This debate will not end between cardiologists, who prefer the patient dry, and Nephrologists who try to avoid AKI in patients who already carries high CV risk.